Eff ect of bimanual sensor glove and unimanual robot-assisted therapy for upper limb function after stroke
Efekt bimanuální senzorické rukavice a unimanuální roboticky asistované terapie na funkci horní končetiny po cévní mozkové příhodě
Úvod: Porucha funkce horní končetiny u pacientů po CMP významně ovlivňuje provádění běžných denních činností (activities of daily living; ADL). Většina ADL je bimanuální, zatímco mnoho konvenčních ergoterapeutických technik je založeno na unimanuálním přístupu. Cílem studie je porovnat dlouhodobé účinky bimanuální a unimanuální roboticky asistované terapie na funkci horní končetiny u pacientů po CMP. Metoda: Pacienti po CMP (n = 40) byli náhodně rozděleni do dvou skupin: roboticky asistovaná bimanuální terapie (BRAT, n = 20) a roboticky asistovaná unimanuální terapie (URAT, n = 20). Terapie trvala 3 týdny a probíhala 5 dní v týdnu, 30 min denně pro obě skupiny. Výsledky intervence byly hodnoceny pomocí Upper Extremity Motor Activity Log (UE MAL) a Motor Assessment Scale (MAS) v časech T0, T1 a T2 (jednoměsíční sledování). K posouzení svalové síly byl použit Motricity Index (MI). Výsledky: BRAT statisticky významně zlepšila funkci horní končetiny v kategorii 7-Hand motion (v T2) a 8-Advantage hand motion (v T1 a T2) dle MAS. Závěr: BRAT má pozitivní vliv na jemnou motoriku a funkci horní končetiny po dokončení terapie a dokonce i po jednoměsíčním sledování. Použití BRAT v kombinaci s konvenční terapií může být účinné při obnově funkce horní končetiny u pacientů po cévní mozkové příhodě se středně těžkou až těžkou hemiparézou.
Klíčová slova:
cévní mozková příhoda – hemiparéza – rehabilitace – senzorická rukavice – běžné denní činnosti – bilaterální roboticky asistovaná terapie
Authors:
K. Hoidekrová 1-3; V. Rogalewicz 1; M. M. Jahromi 4; M. Sobrova 2; D. Pavlů 3
Authors place of work:
Department of Rehabilitation Medicine, First Faculty of Medicine, Charles University and General University Hospital in Prague, Czech Republic
1; Kladruby Rehabilitation Centre, Kladruby u Vlašimi, Czech Republic
2; Department of Physiotherapy, Faculty of Physical Education and Sport, Charles University, Prague, Czech Republic
3; Third Faculty of Medicine, Charles University, Prague, Czech Republic
4
Published in the journal:
Cesk Slov Neurol N 2024; 87(2): 114-121
Category:
Původní práce
doi:
https://doi.org/10.48095/cccsnn2024114
Summary
Introduction: An upper limb functional disability in stroke patients significantly affects activities of daily living (ADL). Most ADL are bimanual, whereas many conventional occupational therapy techniques are based on a unimanual approach. The aim of the study focuses on comparing long--term effects of bimanual and unimanual robot-assisted therapies for upper limb function in stroke patients. Methods: Stroke patients (N = 40) were randomly divided into two groups: robot-assisted bimanual therapy (BRAT, N = 20) and unimanual therapy (URAT, N = 20). Sessions lasted for three weeks running five days a week and 30 min per day for both groups. Outcome measures were the Upper Extremity Motor Activity Log (UE MAL) and the Motor Assessment Scale (MAS) at times T0, T1, and T2 (one month follow-up). Additionally, the Motricity Index (MI) was used to assess force control. Results: BRAT statistically significantly improved upper limb function in category 7-Hand motion (at T2) and 8-Advantage hand motion (at T1 and T2) in MAS. Conclusions: BRAT has a positive effect on fine motor and upper limb function after completion and even after the monthly follow-up. The use of BRAT in combination with conventional therapy appears to be effective in restoring upper limb function in stroke patients with moderate to severe hemiparesis.
Keywords:
stroke – hemiparesis – Activities of daily living – rehabilitation – sensor glove – bilateral robot-assisted training
Introduction
Hemiparesis is one of the most common sensorimotor deficits in 75% of stroke patients [1]. Functional disability of the upper limb is one of the most common deficits after stroke, which is carried into the chronic phase and can affect patients’ participation in activities of daily living (ADL) such as limited grasp ability. Based on the reports, just 5–20% of subjects gain full functional reintegration of the upper limb [2–4]. Moreover, there are indications that up to one third of patients in the acute phase after stroke can achieve full functional recovery of the hand [5].
From a clinical view, recovery of upper limb function and motion is one of the main goals of occupational therapy (OT) in neurorehabilitation due to its importance in inhibiting long-term dependence in ADL, leisure activities, and social and work activities [6].
Robot-assisted therapy has promised to improve the probability of cortical reorganization based on the involvement of the paretic upper limb while transferring the reacquired upper limb function to real ADL [7]. Therefore, it can be considered an adjunctive technique to increase the intensity of treatment which is an important factor for upper limb functional recovery.
Robot-assisted therapy is based on three main fundamental principles aiming to influence brain neuroplasticity as much as possible. First, adequate intensity of therapy alongside with a high number of repetitive movements; second, addressing sensorimotor integration as a crucial principle for motor learning in stroke patients; and third, adequate attention and motivation of the patient during therapy [7]. The use of robot--assisted therapy leads to a deeper immersion in the treatment of patients (e. g., it provides external motivators) to improve their relevance for activities which they undertake in real life (a real task-oriented therapy, a therapy focused on the patient, comprehensive tasks) to improve the feedback strategy (e. g., increase the feedback for fails and optimal results) and also the learning strategy (e. g., to use new control strategies) [7].
Recently, the popularity of robot-assisted devices in paretic hand rehabilitation after stroke increased considerably [8]. Chen et al. showed that using robot-assisted device was feasible and they rated the performance of the patient as well [9], while Chu et al. in their narrative review asserted that the most common device used in studies was a pneumatic actuator to guide finger flexion/extension [8]. Lee et al. observed a higher level of facilitation of the upper limb using the Gloreha device in sub-acute post-stroke patients [10]. Villafañe et al. asserted that it could be a conjunctive therapy technique for spasticity and pain in the paretic hand [11], possibly leading to better paretic function.
However, Chien et al. mentioned the effects of robot-assisted therapy in improving function or disability after chronic stroke are not significantly superior than those of other techniques [12].
Considering all the data about robotic rehabilitation for upper limb function, it can be concluded that there is a wide selection of different devices with contradictory results; more research is needed in different settings.
This article aimed to evaluate the effect of bimanual robot-assisted therapy with a sensor glove and a unimanual robot-assisted approach on upper limb function in post-stroke patients at the end of robotic therapy and at one-month follow-up as most ADLs are bimanual, where coordination of both upper limbs is often required [13].
Materials and methods
Study design
This research is designed as a monocentric randomized controlled parallel two-arm single-blind study. Group A participated in bimanual robot-assisted therapy with the Gloreha Sinfonia (R-Touch Pro) sensor glove (BTL ROBOTICS, Brno, Czech Republic), and group B used unimanual robot-assisted therapy with the Gloreha Sinfonia passive glove. In both groups, robot-assisted therapy was provided for three weeks, five times a week, for 30 min each day, and both groups had 30 min of conventional OT five-times a week. After the end of robot-assisted therapy, both groups continued the same conventional therapies with an intensity of one hour of OT per day, 5 days a week. No other form of robot-assisted therapy for the upper limb was used for one month (between T1 and T2) after the end of Gloreha Sinfonia robot-assisted therapy.
Fifty-six out of a total of 412 patients were assessed for eligibility in the brain injury rehabilitation (BIR) program during the respective period. Sixteen patients did not meet all of the criteria, and 40 patients were included in the study. These 40 participants finished 15 sessions with Gloreha Sinfonia and 36 were also evaluated one month after the last Gloreha robot-assisted therapy session (Fig. 1).
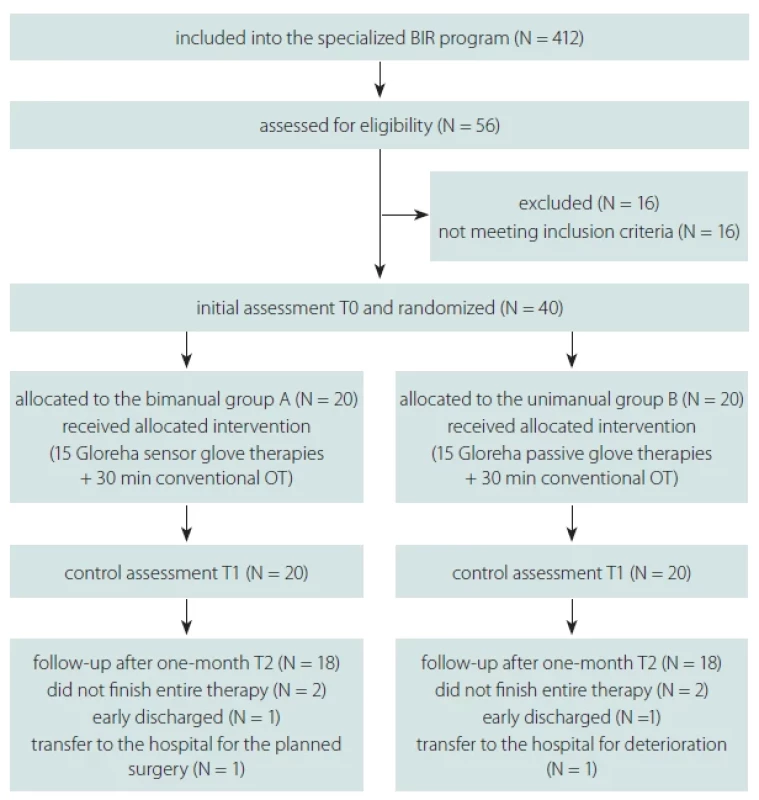
Obr. 1. Vývojový diagram výběru, randomizace a účasti pacientů ve studii.
BIR – rehabilitace poranění mozku; N – počet; OT – ergoterapie
Setting
The study was carried out within the scope of a highly intensive comprehensive BIR program. The BIR program corresponds to the specific needs of stroke patients according to international clinical recommendations for the management of care services for post-stroke patients [14].
The BIR is a multidisciplinary intensive rehabilitation program, which includes one hour of individual OT, one hour of physiotherapy (PT), one hour of speech therapy, and one hour of psychology. The BIR also includes limb positioning, group therapies in OT and PT, and computerized cognitive training. The average time required for comprehensive neurorehabilitation is about 4 to 5 hours per day. The entire program lasts 12 weeks [15].
Participants
Post-stroke patients, who were concurrently included in the rehabilitation BIR program in the Kladruby Rehabilitation Centre, were included in this study. Inclusion criteria were (a) age from 35 to 65 years, (b) no longer than 7 months since the onset of stroke, (c) perform 1–5 activities in the Upper Extremity Motor Activity Log, (d) perform 1–3 activities from the Motor Assessment Scale, and (f) patients included in the BIR program attended at least two out of four disciplines: OT, PT, speech therapy, and psychology. Patients suitable for the study were asked to participate.
Exclusion criteria were comorbidities such as MS, Parkinson‘s disease, rheumatoid arthritis, spinal disease, spatial neglect (diagnosed according to the Catherine Bergego Scale [16] and Bell test [17]), apraxia, severe cognitive deficits, severe phatic disorder (except expressive aphasia), unstable fractures, noncooperative or aggressive behavioral disorders, and severe spasticity according to the modified Ashworth Scale [18] (mAS) > 3.
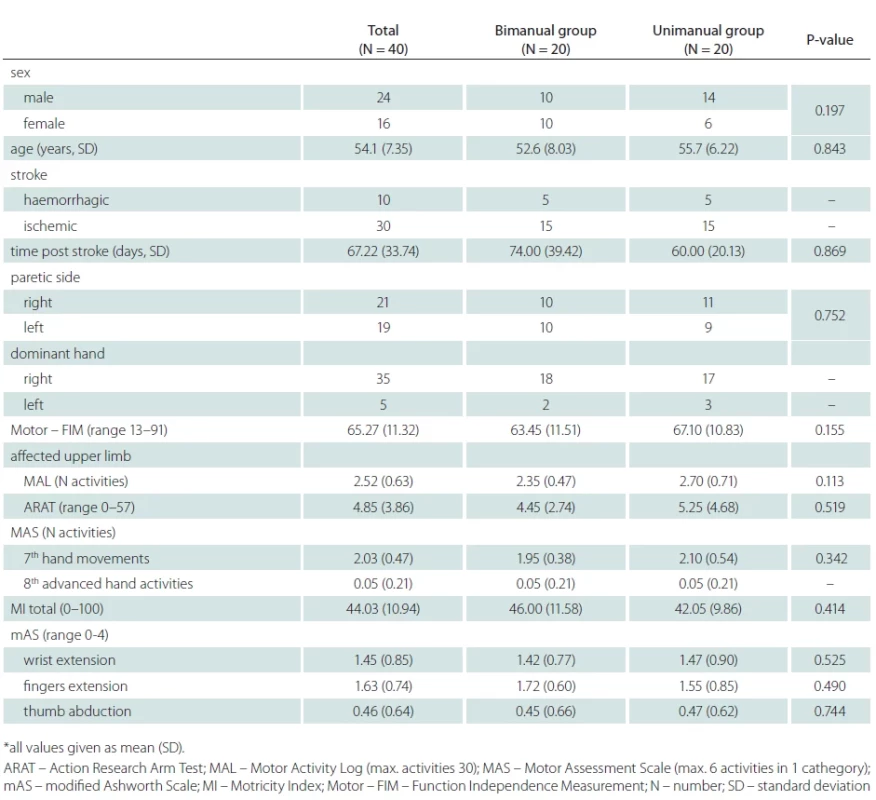
Table 1 shows that there are no significant differences between the demographic characteristics and/or functional status at the time of admittance, as well as paresis severity of the upper limb.
Intervention
During individual OT, robot-assisted Gloreha Sinfonia was applied either as a unimanual therapy or a bimanual therapy (according to random lottery). The Gloreha Sinfonia is a robotic exoskeleton for the neurorehabilitation of the upper limb, which can facilitate the post-stroke patient in all phases of recovery. It can support the movement of the finger joints in passive, active assisted, and active modes, and can be used in unimanual or bimanual approaches. Gloreha Sinfonia consists of a complete set of passive gloves, sensor gloves, braces, accessories for finger mobilization, a dynamic support to compensate the weight of the arm stimulating software equipped with 3D animation, a voice guide, and audio video effects [19].
During unimanual therapy, one robot-assisted Gloreha Sinfonia glove was put on. The glove was equipped with rods for passive mobilization. Therapy consisted of three exercises – (a) making a fist, (b) finger waves, and (c) object grasping. These exercises were chosen intentionally from the Gloreha Sinfonia exercise library, because their content corresponded most to the performance of the exercises in bimanual therapy. Each exercise was combined with action observation therapy lasting seven minutes. Performance velocity of the exercise (flexion and extension) was based on the severity of paresis (or, more precisely, on the spasticity degree evaluated by mAS). During action observation therapy, the patient observed the motion of just one upper limb, which showed and copied the motion of the rods simultaneously.
Bimanual robot-assisted therapy included the use of two gloves. The sensor glove was put on the unaffected hand controlling the second passive glove on the paretic upper limb. During action observation therapy, the patient observed both upper limbs, which showed simultaneous movement of the sensor and passive gloves. The session consisted of three exercises for palm grasping (a fist, grasping, and reaching), which were set for one minute for the action observation therapy itself and for 6 min for mobilization, including action observation therapy with upper limb movements.
Outcome measures
Evaluation methods were chosen from a set of tests that are a standard part of the BIR and are commonly used in post-stroke patients.
All the testing methods outlined below were carried out at baseline (T0), after the 15th session (T1), and one month after the end of the last session of the robot-assisted therapy (T2).
The following tests were used to compare the two therapeutic approaches (unimanual versus bimanual robot-assisted therapies):
The Motor Assessment Scale (MAS) is an evaluation of daily motor function after stroke. For the study, two subcategories were chosen related to the function of the upper limb of the 7th category (hand motion) and 8th category (advanced hand motion). Both categories were evaluated on a seven-point scale (0–6), where the patient received one point for each accomplished task. The maximum score for each category is six points, which indicates optimal motor function for that category [20].
The Upper Extremity Motor Activity Log (UE MAL) is a semi-structured interview questionnaire that evaluates the daily use of the upper limb in post-stroke patients [21]. A modified version of UE MAL consisting of 30 activities was used for the study, with a quantitative evaluation on a scale of 0 or 1 (not done/done) and a complete score of the activities performed.
The Motricity Index (MI) is an ordinal method for measuring the strength of limbs. For our study, the upper limb evaluation was used, which includes three tasks: (a) the proximal joint – abduction in the shoulder joint, (b) the middle joint – flexion in the elbow, and (c) the distal part of the limb, where the patient must grasp and hold a 2.5 × 2.5 cm cube. The score for the proximal and middle joint is: 0 (no movement) /9/14/25/33 (normal strength), while the score for grasp strength is 0 (no movement) /11/19/22/26/33 (normal grasp strength) [22].
Data analysis
The Shapiro-Wilk test was used to determine whether the distribution of the monitored values was normal. It showed that the monitored values were not normally distributed. Therefore, nonparametric tests were chosen to be used in the following calculations. The chi-square test was used for the appraisal of homogeneity of the two groups using basic qualitative demographic parameters (sex, type of stroke, type of hemiparesis), while the Wilcoxon signed-rank test was used to prove the equality of medians in quantitative parameters (age, time from the onset of stroke).
Clinical data of patients in both groups were characterized by their arithmetic means and standard deviations. The chi - -square test (UE MAL, MAS, mAS) and Mann-Whitney test (MI – total score) were used for the comparison of clinical data of the paretic upper limb for patients in both groups. The Mann-Whitney test was also used to compare the cognitive function level evaluated using the Mini Mental States Examination in both groups.
In addition, the Mann-Whitney test was used to compare total scores from UE MAL and MI at times T0–T1 in both groups, while Pearson’s chi-squared test was used for MAS and MI.
Changes in parameters T0–T2 in groups A and B in UE MAL functional tests, the 8th category in MAS, and the total score in MI were compared using the Mann-Whitney test, while Pearson’s chi-squared test was used for the 7th category in MAS. The significance level of 0.05 was chosen for all tests.
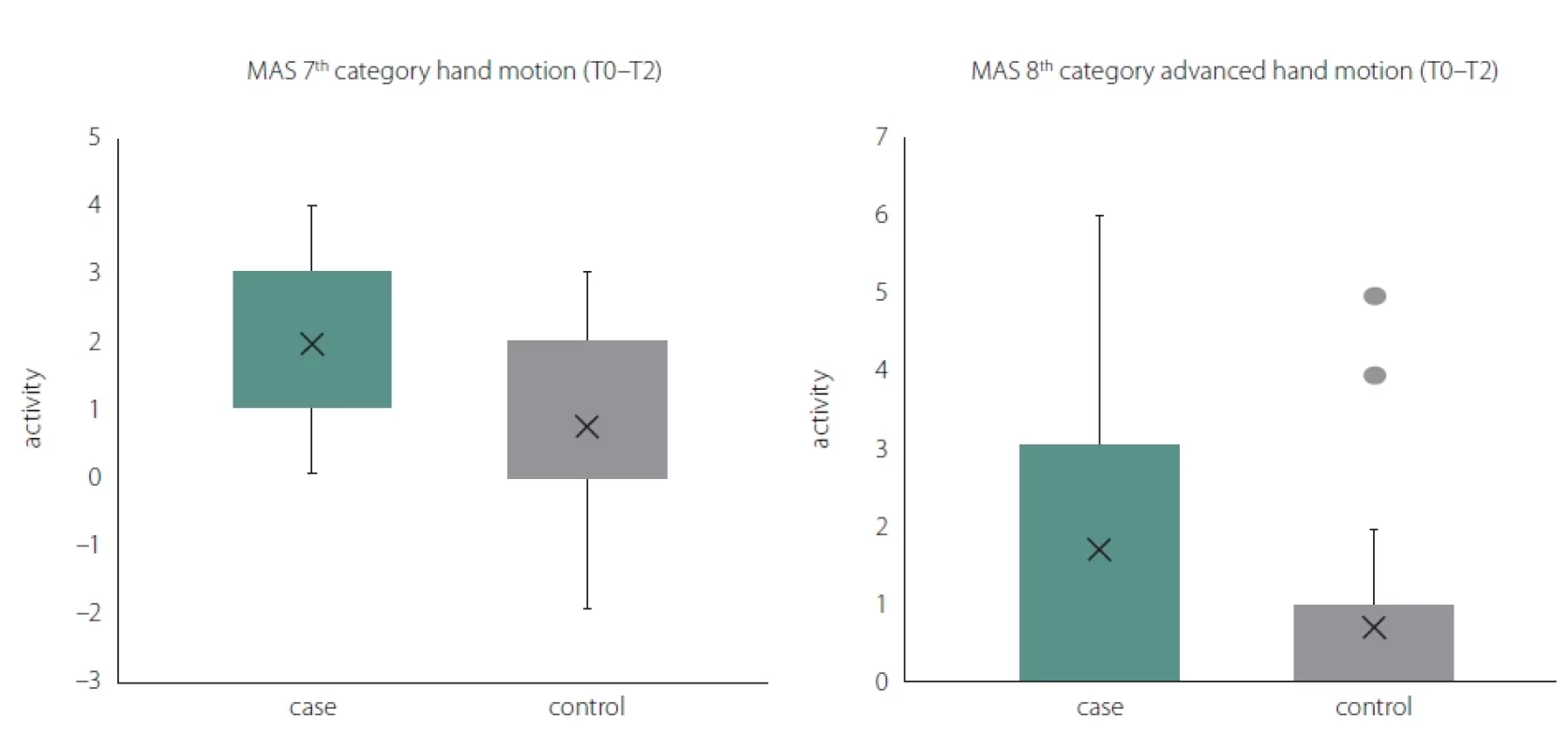
Obr. 2. Krabicové grafy, které ukazují statisticky významné funkční zlepšení dle MAS (T0–T2).
MAS – Motor Asessment Scale
Results
Clinical outcomes
After the last session with the robot-assisted Gloreha glove at T1, a statistically significant difference (P = 0.038) was detected in the MAS 8th category Advanced hand motion. The difference was detected in favor of group A, where the bimanual approach was applied to the functional use of the paretic upper limb.
At one-month follow-up, a statistically significant difference was detected in favor of the group with the bimanual approach in T2 in the MAS 8th category advanced hand motion (P = 0.044) and the MAS 7th category hand motion (P = 0.015) (Fig. 2).
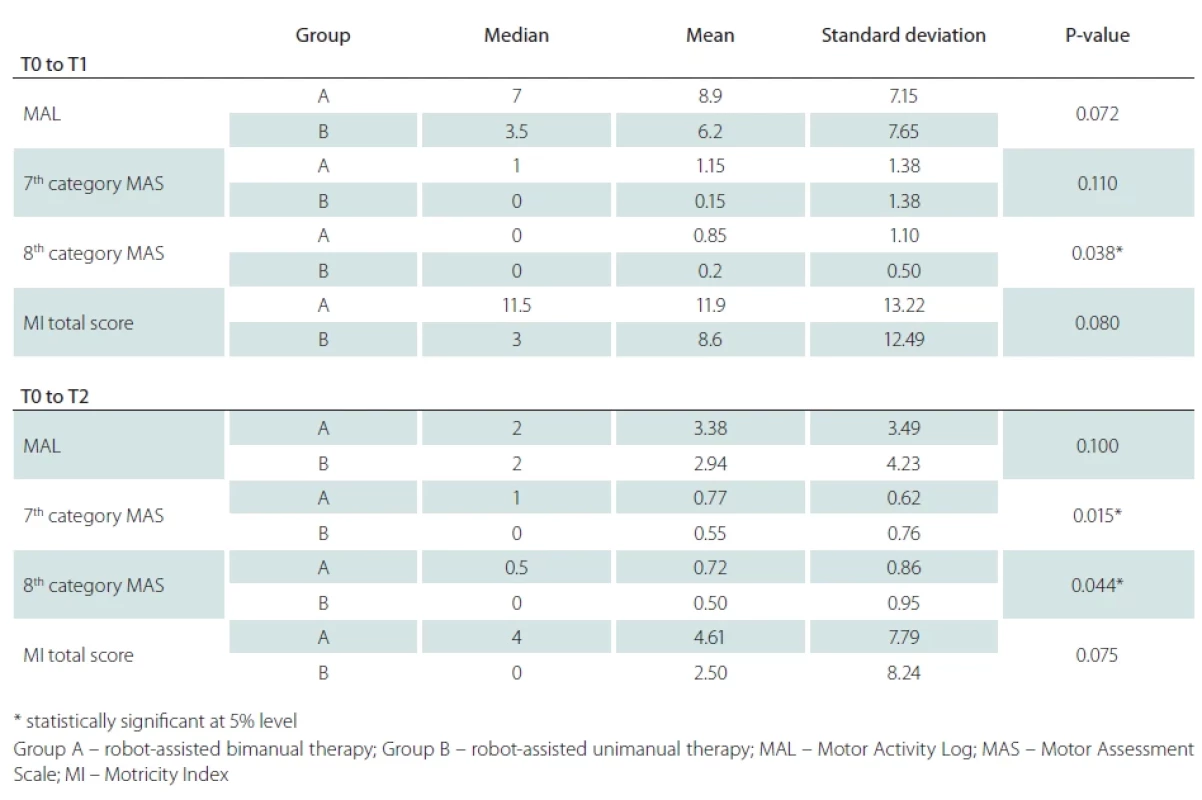
Differences in favor of the group with the bimanual approach in the second functional test UE MAL (P = 0.072 in T1 and P = 0.100 at T2), as well as force control of the total score of MI (P = 0.080) were statistically insignificant (Tab. 2).
Discussion
Our results showed statistically significant changes in the functional evaluation of the upper extremities in MAS at both times in the 8th category. This could show the demand for hand motor control. Moreover, these results were related to the robot-assisted therapy setting, which was focused on the distal upper limb part – the hand.
There are three main reasons for the effectivity of robotic assistive devices for hand rehabilitation. First, it can reduce spasticity so that movement is facilitated. Second, it is much easier for patients to perform the exercise by improving somatosensory input resulting in better motor planning and faster recovery. Last but not least, passive range of motion exercises can reduce inhibition in affected brain areas [23].
Statistically significant results were detected in the seventh category only at time T2 (P = 0.015). The main differences between MAS and UE MAL were the working positions and the manner in which the tasks were performed. Compared to this, the 8th category in MAS contains only one of six activities, where shoulder flexion above 90° combined with external rotation is required. The other activities are tested on a table. It could be hypothesized that muscle weakness, pain, or joint luxation may greatly reduce functional usage of the paretic upper limb. This theory is supported by Blennerhassett et al. [24], who predicted a 64% probability of an increase in the incidence of shoulder pain with every point lost in the sixth category for MAS (the arm activity). Wu et al. [25] even suggested including a bimanual evaluation of ADL (e. g., the ABIL--HAND questionnaire). This could better reflect upper limb bimanual usage, which corresponded more to bimanual robot-assisted therapy settings.
In accordance to the results of this study, Yuan et al. [26], reported that the effects of bimanual therapy were greater than unilateral mode in stroke patients. Although they used another device and other outcome measurements, they explained that higher priority of bimanual compared to unilateral therapy can possibly be related to better inter-hemispheric and intra-hemispheric connections resulting in activation of the ipsilesional primary motor cortex and supplementary motor area. This, finally, can lead to rebalancing interhemispheric inhibition caused by stroke.
The recorded presence of pain in the shoulder, elbow, or hand could be caused by the application of UE MAL. Some activities in the test are performed above the shoulder horizontal line or with the individual leaning forward. These are the positions in which the pain could be accented. Patients complaining of upper extremity pain were evenly distributed between the two groups (A = 7, B = 8). Similar subjective pain perception was described in probands from both groups; it was described in marginal shoulder positions or during passive stretching of the shoulder over 90° in abduction.
The results of MI in the upper extremities differ in many studies on Gloreha robot-assisted therapy, as well as in our study. The results are affected by the study design and sample size. Based on previous studies [27,28], there was no statistically significant improvement in muscle strength after a three-week period of Gloreha therapy (two therapy sessions per week), while in another study, a statistically significant improvement was detected in the total score after a two-month period of Gloreha therapy and even two months after the final therapy session. Other studies [26,27] did not correspond to each other in muscle strength results. Vanoglio et al. [30] mentioned significant improvement in the total score for the upper limb after a six-week therapy, while Montecchi et al. [29] reported no improvement after a one-week therapy. Our results and complete results of prior studies can help with their interpretation.
Previously published studies [27,30] claim that the amount and intensity of robot-assisted therapy can influence the effect of therapy with regard to limb muscle strength. The designs of those studies were different compared to groups with different robot-assisted interventions. Bernocchi et al. [28] evaluated, by means of MI, the long-term effects of therapy on muscle strength (P = 0.0371), but there was no comparison with a control group. However, regardless of muscle strength, in a recent systematic review and meta-analysis, it is shown that the most positive results of using robotic devices are immediate and evidence for long-term effects is insufficient [31].
Improvement (both statistical and clinical) can also be detected by MI in proximal parts of the limb for single areas (shoulder, elbow, hand), although robot-assisted therapy focused on the hand. This result is supported by the study by Krebs et al. [32], who applied robot-assisted therapy on distal parts, but the improvements were also detected in the proximal parts of the limb.
Although statistically significant results are usually presented at the significance level of 0.05, results that are significant at the level of 0.10 are also important for this study. The study includes two rather small groups of 20 probands. The results were closer to the significance level of 0.05 when 40 probands were included, which is an important piece of information for future studies. However, it is mentioned that using Gloreha gloves can save time for therapists as it is known to be a safe device that also can provide feedback for patients [11], which could be another possible explanation for their improvements in this study. Considering the feedback effect, it is possible that using robotic rehabilitation combined with action observation can increase attention, confidence, and motivation to exercise [33,34]. In line with this theory, it is mentioned that robotic rehabilitation can affect cognitive abilities like neglect after stroke [35]. To our knowledge, if we improve cognition and give the patient better feedback regarding the treatment process/steps, it can possibly lead to better motor function, and finally improve limb movements within ADL.
Limitations
As for the first limitation, we have to mention the limited size of the sample. In many cases, this was probably the reason why sufficient statistical significance was not confirmed (see P-values between 0.05 and 0.10).
The study was also affected by occasional technical difficulties with the Gloreha Sinfonia glove. Most often, the difficulty was in releasing the mobilization rod from the main piston or releasing the pistons. The device was always quickly fixed, so that the continuity of the therapy sessions was never disturbed by such technical difficulties.
Another problem that appeared during the investigation was an incorrect recording of completed cycles in bimanual robot-assisted therapy. Completion of cycles was recorded in only 753 cases (exercises). The incorrect recording was probably caused by an acceleration of the motion of the sensor glove on the healthy limb. However, simultaneous movements of the sensor and passive gloves were set up in the basic setting, and the passive glove moved slightly slower than the sensor glove.
Conclusions
The study reflects the current situation in robot-assisted therapy and neurorehabilitation through the application of Gloreha Sinfonia new sensor glove. This study combined conventional methods with robotic therapy and focused on the function and functional skills of the paretic upper limb in stroke patients. At the same time, it reflected on the most common ADL activities and explored a bimanual robot-assisted approach.
We showed that using bimanual robotic therapy with a sensor glove positively affected upper limb function as compared to unimanual robotic therapy in stroke patients. Although both groups received the same conventional OT after the end of robot-assisted therapy, there was statistically significant improvement in the robot-assisted bimanual therapy group after the end of whole therapy, and even one month after the end of the application of the robotic glove. The results suggest that robot-assisted bimanual therapy could influence the recovery of upper limb function more than long-term robot-assisted unimanual therapy.
Clinical implications
Based on this study, it can be concluded that the combination of conventional OT and bimanual robot-assisted therapy could have long-term effects on the recovery of upper limb function in patients after stroke. Further studies with a larger sample size are needed to address the long-term effects of robot - -assisted therapy on upper limb function and self-sufficiency in stroke patients.
Ethical aspects
The study was conducted in compliance with the 1975 Declaration of Helsinki (and its 2004 and 2008 revisions), and approved by the Ethics Committee of Rehabilitation Center Kladruby (reference number of 2017/14, date of approval January 1, 2018) and Ethical Committee of the Faculty of Physical Education and Sport, Charles University with a reference number of 221/2017 and date of approval January 29, 2018. Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the patient (s) to publish this paper.
Conflict of interest
The authors declare they have no potential conflicts of interest concerning drugs, products, or services used in the study.
Zdroje
1. Trlep M, Mihelj M, Puh U et al. Rehabilitation robot with patient-cooperative control for bimanual training of hemiparetic subjects. Advanced Robotics 2011; 25 : 1949–1968. doi: 10.1163/016918611X588853.
2. Calabrò RS, Accorinti M, Porcari B et al. Does hand robotic rehabilitation improve motor function by rebalancing interhemispheric connectivity after chronic stroke? Encouraging data from a randomised-clinical-trial. Clin Neurophys 2019; 130 (5): 767–780. doi: 10.1016/j.clinph.2019.02.013.
3. Cruz EG, Waldinger HC, Kamper DG. Kinetic and kinematic workspaces of the index finger following stroke. Brain 2005; 128 (Pt 5): 1112–1121. doi: 10.1093/brain/awh432.
4. Jan S, Arsh A, Darain H et al. A randomized control trial comparing the effects of motor relearning programme and mirror therapy for improving upper limb motor functions in stroke patients. J Pak Med Assoc 2019; 69 (9): 1242–1245.
5. Beebe JA, Lang CE. Active range of motion predicts upper extremity function 3 months after stroke. Stroke 2009; 40 (5): 1772–1779. doi: 10.1161/STROKEAHA.108. 536763.
6. Coupar F, Pollock A, van Wijck F et al. Simultaneous bilateral training for improving arm function after stroke. Cochrane Database Syst Rev 2010; 2010 (4): CD006432. doi: 10.1002/14651858.CD006432.pub2.
7. Johnson MJ. Recent trends in robot-assisted therapy environments to improve real-life functional performance after stroke. J Neuroeng Rehabil 2006; 3 : 29. doi: 10.1186/1743-0003-3-29.
8. Chu C-Y, Patterson RM. Soft robotic devices for hand rehabilitation and assistance: a narrative review. J Neuroeng Rehabil 2018; 15 (1): 9. doi: 10.1186/s12984-018-0350-6.
9. Chen Y-M, Lai S-S, Pei Y-C et al. Development of a novel task-oriented rehabilitation program using a bimanual exoskeleton robotic hand. J Vis Exp 2020; 159. doi: 10.3791/61057.
10. Lee H-C, Kuo F-L, Lin Y-N et al. Effects of robot-assisted rehabilitation on hand function of people with stroke: a randomized, crossover-controlled, assessor-blinded study. Am J Occup Ther 2021; 75 (1): 7501205020p1–7501205020p11. doi: 10.5014/ajot.2021. 038232.
11. Villafañe JH, Taveggia G, Galeri S et al. Efficacy of short-term robot-assisted rehabilitation in patients with hand paralysis after stroke: a randomized clinical trial. Hand 2018; 13 (1): 95–102. doi: 10.1177/1558944717692096.
12. Chien W, Chong Y, Tse M et al. Robot-assisted therapy for upper-limb rehabilitation in subacute stroke patients: a systematic review and meta-analysis. Brain Behav 2020; 10 (8): e01742. doi: 10.1002/brb3.1742.
13. Cauraugh JH, Lodha N, Naik SK et al. Bilateral movement training and stroke motor recovery progress: a structured review and meta-analysis. Hum Mov Sci 2010; 29 (5): 853–870. doi: 10.1016/j.humov.2009.09.004.
14. National Stroke Foundation. Clinical guidelines for stroke management 2010. [online]. Available from: https: //extranet.who.int/ncdccs/Data/AUS_D1_Clinical%20Guidelines%20for%20Stroke%20Manage - ment.pdf.
15. Vilimovsky T, Chen P, Hoidekrova K et al. Prism adaptation treatment to address spatial neglect in an intensive rehabilitation program: a randomized pilot and feasibility trial. PLoS One 2021; 16 (1): e0245425. doi: 10.1371/journal.pone.0245425.
16. Bergego C, Azouvi P, Samuel C et al. Functional consequences of unilateral neglect: validation of an evaluation scale, the CB scale. Ann Med Phys Readaptation 1995; 38 : 183–189. doi: 10.1016/0168-6054 (96) 89317-2.
17. Gauthier L, Dehaut F, Joanette Y. The Bells Test: a quantitative and qualitative test for visual neglect. Int J Clin Neuropsychol 1989; 11 (2): 49–54.
18. Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Physical therapy 1987; 67 (2): 206–207. doi: 10.1093/ptj/67.2.206.
19. Bressi F, Santacaterina F, Cricenti L et al. Robotic-assisted hand therapy with gloreha sinfonia for the improvement of hand function after pediatric stroke: a case report. Appl Sci 2022; 12 (9): 4206. doi: 10.3390/ app12094206.
20. Carr JH, Shepherd RB, Nordholm L et al. Investigation of a new motor assessment scale for stroke patients. Phys Ther 1985; 65 (2): 175–180. doi: 10.1093/ptj/65.2.175.
21. Chen H, Lin K, Hsieh Y et al. A study of predictive validity, responsiveness, and minimal clinically important difference of arm accelerometer in real-world activity of patients with chronic stroke. Clin Rehabil 2018; 32 (1): 75–83. doi: 10.1177/0269215517712042.
22. Collin C, Wade D. Assessing motor impairment after stroke: a pilot reliability study. J Neurol Neurosurg Psychiatry 1990; 53 (7): 576–579. doi: 10.1136/jnnp.53.7.576.
23. Hsu C-Y, Wu C-M, Huang C-C et al. Feasibility and potential effects of robot-assisted passive range of motion training in combination with conventional rehabilitation on hand function in patients with chronic stroke. J Rehabil Med 2022; 54: jrm00323. doi: 10.2340/jrm.v54. 1407.
24. Blennerhassett JM, Gyngell K, Crean R. Reduced active control and passive range at the shoulder increase risk of shoulder pain during inpatient rehabilitation post-stroke: an observational study. J Physiother 2010; 56 (3): 195–199. doi: 10.1016/S1836-9553 (10) 70025-4.
25. Wu C, Yang C, Chuang L et al. Effect of therapist-based versus robot-assisted bilateral arm training on motor control, functional performance, and quality of life after chronic stroke: a clinical trial. Phys Ther 2012; 92 (8): 1006–1016. doi: 10.2522/ptj.20110282.
26. Yuan R, Qiao X, Tang C et al. Effects of uni - vs. bilateral upper limb robot-assisted rehabilitation on motor function, activities of daily living, and electromyography in hemiplegic stroke: a single-blinded three-arm randomized controlled trial. J Clin Med 2023; 12 (8): 2950. doi: 10.3390/jcm12082950.
27. Bissolotti L, Villafañe JH, Gaffurini P et al. Changes in skeletal muscle perfusion and spasticity in patients with poststroke hemiparesis treated by robotic assistance (Gloreha) of the hand. J Phys Ther Sci 2016; 28 (3): 769–773. doi: 10.1589/jpts.28.769.
28. Bernocchi P, Mulè C, Vanoglio F et al. Home-based hand rehabilitation with a robotic glove in hemiplegic patients after stroke: a pilot feasibility study. Top Stroke Rehabil 2018; 25 (2): 114–119. doi: 10.1080/10749 357.2017.1389021.
29. Giulia M, Francesca M, Simone T et al. Is passive mobilization robot-assisted therapy effective in upper limb motor recovery in patients with acquired brain injury? A randomized crossover trial. Int J Phys Ther Rehabil 2016; 2 (2): 114. doi: 10.15344/2455-7498/2016/114.
30. Vanoglio F, Bernocchi P, Mulè C et al. Feasibility and efficacy of a robotic device for hand rehabilitation in hemiplegic stroke patients: a randomized pilot controlled study. Clin Rehabil 2017; 31 (3): 351–360. doi: 10.1177/0269215516642606.
31. Zhang L, Jia G, Ma J et al. Short and long-term effects of robot-assisted therapy on upper limb motor function and activity of daily living in patients post-stroke: a meta-analysis of randomized controlled trials. J Neuroeng Rehabil 2022; 19 (1): 76. doi: 10.1186/s12984-022-01058-8.
32. Krebs HI, Volpe BT, Williams D et al. Robot-aided neurorehabilitation: a robot for wrist rehabilitation. IEEE Trans Neural Syst Rehabil Eng 2007; 15 (3): 327–335. doi: 10.1109/TNSRE.2007.903899.
33. Moulaei K, Bahaadinbeigy K, Haghdoostd AA et al. Overview of the role of robots in upper limb disabilities rehabilitation: a scoping review. Arch Public Health 2023; 81 (1): 84. doi: 10.1186/s13690-023-01100-8.
34. Choi J-B, Yang S-W, Ma S-R. The effect of action observation combined with motor imagery training on upper extremity function and corticospinal excitability in stroke patients: a randomized controlled trial. Int J Environ Res Public Health 2022; 19 (19): 12048. doi: 10.3390/ijerph191912048.
35. Bressi F, Cricenti L, Campagnola B et al. Effects of robotic upper limb treatment after stroke on cognitive patterns: a systematic review. NeuroRehabilitation 2022; 51 (4): 541–558. doi: 10.3233/NRE-220149.
Štítky
Dětská neurologie Neurochirurgie NeurologieČlánek vyšel v časopise
Česká a slovenská neurologie a neurochirurgie

2024 Číslo 2
-
Všechny články tohoto čísla
- Stroke-heart syndrome – cardiac complications in ischemic stroke patients
- Experimental model of intracranial aneurysms
- Eff ect of bimanual sensor glove and unimanual robot-assisted therapy for upper limb function after stroke
- Course of myelin oligodendrocyte glycoprotein-associated disease (MOGAD) – a cohort of patients
- Dementia knowledge and attitudes from Third-age university participants
- Factors influencing the outcome in patients with cauda equina syndrome
- Surgical treatment of spontaneous intracranial hypotension
- An unusual occupational accident – a case of penetrating head injury
- Migration of the Kirschner‘s wire into the spine as a cause of long-lasting neurological problems
- Profesor Pavel Kalvach a jeho 80. narozeniny
- Takanori Fukushima
- Komentář k článku autorů Hejčl et al. Chirurgická léčba recidivy intrakraniálních aneuryzmat po ošetř ení klipem
- Hypoglycemia as a complication of diabetes mellitus treatment acute eff ect on central nervous system function
- Neurosurgical interventions during pregnancy
- Česká a slovenská neurologie a neurochirurgie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle
- Course of myelin oligodendrocyte glycoprotein-associated disease (MOGAD) – a cohort of patients
- Factors influencing the outcome in patients with cauda equina syndrome
- Neurosurgical interventions during pregnancy
- Eff ect of bimanual sensor glove and unimanual robot-assisted therapy for upper limb function after stroke
