Dural-based posterior fossa giant cavernous hemangioma masquerading as hemangiopericytoma
Gigantický kavernózní hemangiom zadní jámy nasedající na duru napodobující hemangiopericytom
Autoři deklarují, že v souvislosti s předmětem studie nemají žádné komerční zájmy.
Redakční rada potvrzuje, že rukopis práce splnil ICMJE kritéria pro publikace zasílané do biomedicínských časopisů.
Authors:
G. Dutta 1; D. Sachdeva 1; D. Singh 1; R. Saran 2; A. Reddy 1; I. Mohd 1; M. Garg 1
Authors‘ workplace:
Department of Neuro-Surgery, Govind Ballav Pant Institute of Postgraduate Medical Education and Research (GIPMER), New Delhi, India
1; Department of Pathology, Govind Ballav Pant Institute of Postgraduate Medical Education and Research (GIPMER), New Delhi, India
2
Published in:
Cesk Slov Neurol N 2018; 81(1): 95-97
Category:
Letter to Editor
doi:
https://doi.org/doi: 10.14735/amcsnn201895
Overview
Autoři deklarují, že v souvislosti s předmětem studie nemají žádné komerční zájmy.
Redakční rada potvrzuje, že rukopis práce splnil ICMJE kritéria pro publikace zasílané do biomedicínských časopisů.
Dear editor,
Intracranial cavernous hemangiomas are one of the rarest among all vascular malformations, with a necropsy incidence between 0.39 and 0.53% [1] accounting for 10 – 20% [2] of all vascular malformations. They are usually found in a supratentorial location with 20.7% incidence in the infratentorial region but rarely in the region of cerebellum (6.1%) [3]. The rarity of the cavernous hemangioma in the region of the cerebellum may result in a frequent misdiagnosis thus making the treatment quite challenging. Giant cavernous hemangiomas (GCH) are exceedingly rare, imaging appearance is quite variable and because of slow perfusion, contrast enhancement is not regarded as a characteristic imaging finding of the cavernous hemangiomas, so straightforward diagnosis may again pose a significant challenge [4]. Some of the GCHs may have features similar to those of typical cavernous hemangiomas, while some may be purely cystic, and some may present with significant contrast enhancement along with a mass effect, mimicking neoplasms [5]. We present our experience with a case of GCH that mimicked the appearance of a hemangiopericytoma and discuss its radiological and histopathological findings.
A 25-year-old female patient was admitted to our hospital with a history of persistent occipital headache with intermittent vomiting and unsteadiness of gait during the past 3 months. On examination, she was conscious and oriented. Cerebellar signs were positive on the right side. There were no cranial nerve palsies and ophthalmoscopic examination was normal. Contrast-enhanced computed tomography (CECT) of the brain was suggestive of heterogeneously enhancing extra-axial mass lesion at the right posterior fossa with destruction of the overlying bone causing compression of the 4th ventricle and hydrocephalus. Magnetic resonance imaging (MRI) of the brain revealed a large, well-defined extra-axial mass lesion in the posterior fossa causing compression of the right cerebellum which was T1-isointense, T2/ FLAIR-hyperintense with intensive post-contrast enhancement with necrotic areas and flow voids. There was dural enhancement and destruction of the right occipital bone along with cerebellar tonsillar herniation and obstructive hydrocephalus. There was non-visualization of the right transverse sinus suggestive of compression (Fig. 1). In view of the possible diagnosis of hemangiopericytoma, a retromastoid retrosigmoid approach was used to operate the patient in the sitting position. Intraoperatively, there was a lesion sized 5 × 5.5 cm in the right posterior fossa which was reddish-brown in color, firm and highly vascular with a well-defined plane of cleavage. The dura was hypertrophied, and the overlying bone was eroded by the tumor. Total excision of the lesion was done.
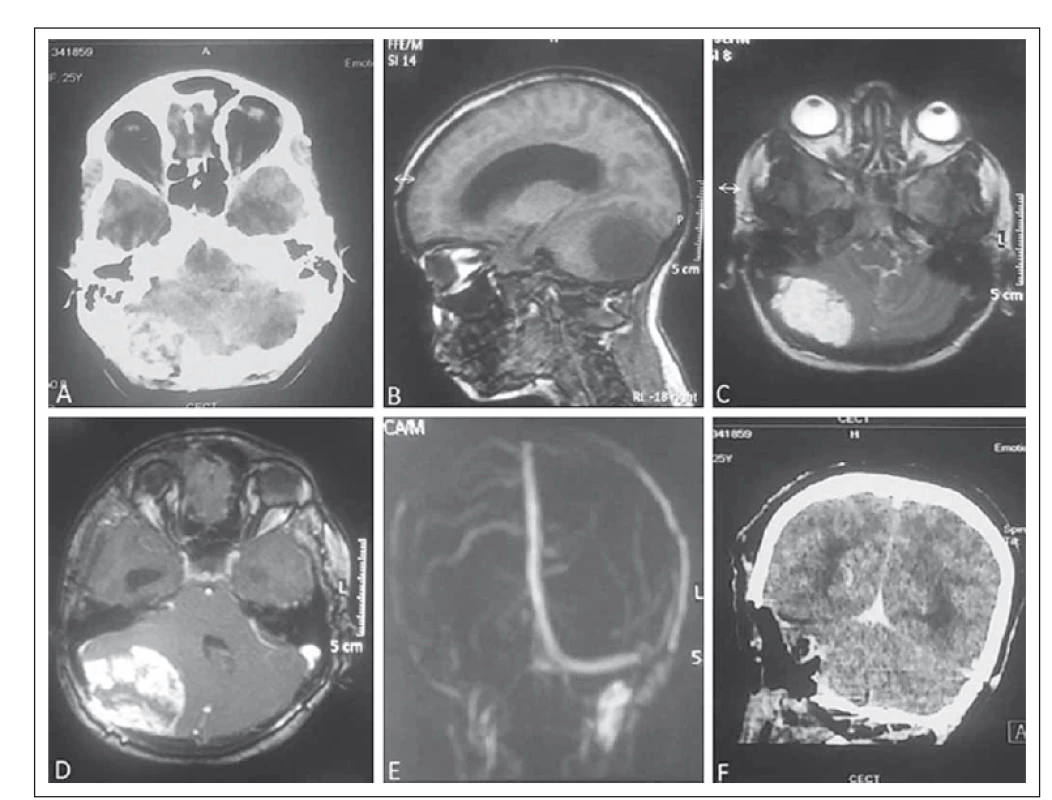
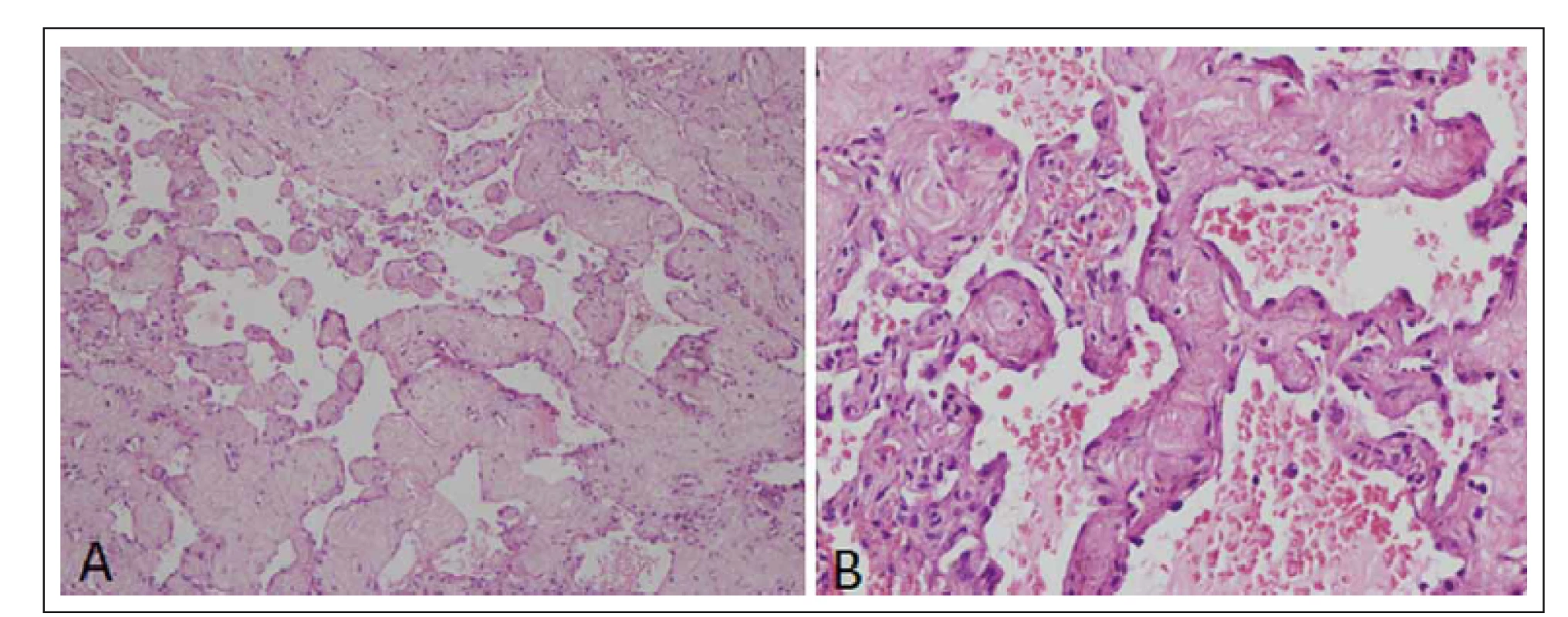
Histopathological examination of the specimen revealed a vascular lesion composed of a variable sized thin-walled vascular channel with dilated and blood filled lumina. Intervening stroma showed delicate collagenization. The lining cells were negative for EMA and S-100 (Fig. 2), was consistent with the diagnosis of cavernous hemangioma.
Postoperatively, the patient did well and was discharged on the 8th post-operative day. A repeat CECT of the brain showed that the lesion was completely removed. At the 3-month follow-up, she continued to do well with a complete resolution of pre-operative symptoms and cerebellar signs.
Cavernous hemangiomas are benign vascular lesions comprised of enlarged, clustered, sinusoidal vessels without intervening parenchymal tissue lined with epithelium. They are most frequently found in the brain parenchyma but can also arise in the spinal cord or in an extra-axial location [6]. The most common presenting symptoms are seizures (37%), hemorrhage (36%), headache (23%), and focal neurological deficit (22%); however, unlike parenchymal lesions, dural-based cavernous hemangiomas commonly present with headache, rather than seizures or hemorrhage [7]. Histologically, they appear as a cluster of hyalinized sinusoidal spaces without neural tissue which differentiates them from telangiectasias [6]. The mean size of cavernous hemangiomas reported in several large series varies from 15 to 19 mm in diameter and according to some authors, the larger the lesion, the higher the chances of symptom recurrence and GCHs were rarely seen [8].
Dural-based cavernous malformations mimicking meningiomas or other neoplastic processes have been reported in the literature and differential diagnosis of such lesions is wide, including hemangiopericytomas, hemangioblastomas, solitary fibrous tumors, dural-based metastases, and angiomatous meningiomas. These lesions tend to beiso - to hypointense on T1-weighted images,mixed to hyperintense on T2-weighted ima-ges, contrast enhancing, and hyperdense on CT [9]. However, these findings are not typical for cavernomas and can also be seen in hemangiopericytomas or meningiomas, thereby clouding the differential diagnosis based on history and radiologic analysis. Several authors have reported that lytic destruction of the adjacent skull on plain X-rays or CT suggests an hemangiopericytoma while hyperostosis suggests a meningioma [10]. Based on this, our pre-operative diagnosis was a hemangiopericytoma; however, the case turned out to be a GCH thus proving the fact that dural-base cavernous hemangiomas may mimic the clinical and radiological features of a hemangiopericytoma. This unique case strengthens the fact that lesions at the posterior fossa may cause diagnostic dilemmas and etiology of such tumors may not always be straightforward. The case also urges to keep cavernous hemangioma as a differential diagnosis of extra-axial lesions at the posterior fossa.
The lack of definitive radiologic and clinical entities differentiating cavernous hemangiomas from other neoplastic lesions can lead to unexpected findings in the operating room and may alter treatment plans especially in cases of hemangiopericytoma, as they are considered to be aggressive lesions. Given the lack of differentiable factors, it is very important for the neurosurgeon to keep in mind the possibility of dural-based cavernous hemangiomas when formulating a differential diagnosis and choosing the appropriate excision technique and postoperative treatment plan. These lesions do not tend to recur in contrast to hemangiopericytomas and can safely be monitored without adjunct therapy.
The unique nature of our case of GCH stems from its size, location in the posterior fossa, dural attachment and appearance similar to hemangiopericytoma. To the best of our knowledge, this has not been described in the literature. Although hemangiomas are benign entities, our patient‘s lesion was in the posterior fossa causing compression, hydrocephalus and cerebellar symptoms that necessitated resection. We would like to encourage others to consider the possibility of hemangioma in the differential diagnosis of dural-based posterior fossa lesions.
The authors declare they have no potential conflicts of interest concerning drugs, products, or services used in the study.
The Editorial Board declares that the manuscript met the ICMJE “uniform requirements” for biomedical papers.
Accepted for review: 20. 4. 2017
Accepted for print: 21. 11. 2017
Dr. Gautam Dutta
Department of Neuro-Surgery, Govind Ballav Pant Institute of Postgraduate Medical Education and Research (GIPMER), New Delhi, India
1, Jawaharlal Nehru Marg
Delhi 110002, India
e-mail: gautamblue@hotmail.com
Sources
1. Otten P, Pizzolato GP, Rilliet B et al. A propos de 131 cas d’angiomes caverneux (cavernomas) du système nerveux central repérés par l’analyse rétrospective de 24 535 autopsies. Neurochirurgie 1989; 35(2): 82 – 83.
2. Deshmukh VR, Albuquerque FC, Zabramski JM et al. Surgical management of cavernous malformation involving the cranial nerves. Neurosurgery 2003; 53(2): 352 – 357.
3. Kadota O, Sakaki S, Kumon Y et al. Large cystic cavernous angioma of the cerebellum. Neurol Med Chir (Tokyo) 1994; 34(11): 768 – 772.
4. Thiex R, Krüger R, Friese S et al. Giant cavernoma of the brain stem: value of delayed MR imaging after contrast injection. Eur Radiol 2003; 13 (Suppl 6): L219 – L225.
5. Gelal F, Feran H, Rezanko T et al. Giant cavernous angioma of the temporal lobe: a case report and review of the literature. Acta Radiol 2005; 46(3): 310 – 313.
6. Simard MJ, Garcia-Bengochea F, Ballinger WE Jr et al. Cavernous angioma: a review of 126 collected and 12 new clinical cases. Neurosurgery 1986; 18(2): 162 – 172.
7. Lewis AI, Tew JM Jr, Payner TD et al. Dural cavernous angiomas outside the middle cranial fossa: a report of two cases. Neurosurgery 1994; 35(3): 498 – 504.
8. Curling OD Jr, Kelly DL Jr, Elster AD et al. An analysis of natural history of cavernous angiomas. J Neurosurg 1991; 75(5): 702 – 708. doi: 10.3171/ jns.1991.75.5.0702.
9. Vogler R, Castillo M. Dural cavernous angioma: MR features. AJNR Am J Neuroradiol 1995; 16(4): 773 – 775.
10. Tashjian VS, Khanlou N, Vinters HV et al. Hemangiopericytoma of the cerebellopontine angle: a case report and review of the literature. Surg Neurol 2009; 72(3): 290 – 295. doi: 10.1016/ j.surneu.2008.06.016.
Labels
Paediatric neurology Neurosurgery NeurologyArticle was published in
Czech and Slovak Neurology and Neurosurgery

2018 Issue 1
-
All articles in this issue
- Injury as a cause of extrapyramidal syndrome
- Injury as a cause of extrapyramidal syndromes
- Injury as a cause of extrapyramidal syndromes Comment on controversies
- Olfactory groove meningiomas – surgical treatment, surgical risks and sense of smell preservation
- Neuropalliative and rehabilitative care in patients with an advanced stage of progressive neurological diseases
- Protective factors for cognitive impairment in multiple sclerosis
- Assessment of cognitive functions using short repeatable neuropsychological batteries
- Test of gestures (TEGEST) for a brief examination of episodic memory in mild cognitive impairment
- The importance of morphological and clinical classifications of lumbar spine stenosis in the preoperative planning
- Parosmia and phantosmia in patients with olfactory dysfunction
- SCN1A mutation positive Dravet syndrome, genetic aspects and clinical experiences
- Sentence comprehension in Slovak-speaking patients with Parkinson disease
- The pilot study of effect of outpatient functional electrical stimulation of peroneal nerve
- A neurological view on spondylodiscitis
- Statins and their effects on the peripheral nervous system
- Cavernous sinus thrombosis – still occurring complication of rhinosinusitis
- T1 radiculopathy due to massive disc herniation at T1/2
- Neuropathological post-mortem examination of the brain and the spinal cord in ten key points – What can a neurologist expect from the neuropathologist’s confirmation of the clinical diagnosis in neurodegenerative diseases?
- Long-term follow up of a patient with primary cervical spinal cord meningeal melanocytoma
- Neurosurgical resident training in the Czech Republic
- Alternative forms parallel to the Czech versions of Rey Auditory Verbal Learning Test, Complex Figure Test and Verbal Fluency
- Leiomyoma of the palm
- Dural-based posterior fossa giant cavernous hemangioma masquerading as hemangiopericytoma
- Czech and Slovak Neurology and Neurosurgery
- Journal archive
- Current issue
- About the journal
Most read in this issue
- A neurological view on spondylodiscitis
- Parosmia and phantosmia in patients with olfactory dysfunction
- Assessment of cognitive functions using short repeatable neuropsychological batteries
- Cavernous sinus thrombosis – still occurring complication of rhinosinusitis
