Equivalence of Montreal Cognitive Assessment alternate forms
Ekvivalence alternativních verzí Montrealského kognitivního testu
Cíl: Cílem studie je popis psychometrických vlastností standardní verze Montrealského kognitivního testu (MoCA-SV) a dvou alternativních forem (MoCA-AV). V české verzi je nedostatek informací o test-retestovém efektu a reliabilitě MoCA.
Metodika: Sedmdesát kognitivně zdravých osob (průměrný věk 50,33 ± 26,47) bylo vyšetřeno v jednom sezení pomocí MoCA-SV a MoCA-AV (7.2 a 7.3) ve znáhodněném pořadí.
Výsledky: Mezi MoCA-SV a MoCA-AV 7.3 neexistuje statisticky významný rozdíl v celkovém hrubém skóru, zatímco v MoCA-AV 7.2 je obtížnější verze testu ve srovnání s MoCA-SV a MoCA-AV 7.3. Hlubší analýza odhalila další rozdíly mezi subtesty verzí MoCA. Celkové skóry MoCA však ve všech verzích naznačují přijatelné hodnoty konvergentní a diskriminační validity.
Závěr: Naše data naznačují, že MoCA-SV i MoCA-AV 7.3 jsou ekvivalentní a užitečné pro opakovanou administraci a minimalizaci test-retestového efektu.
Autoři deklarují, že v souvislosti s předmětem studie nemají žádné komerční zájmy.
Redakční rada potvrzuje, že rukopis práce splnil ICMJE kritéria pro publikace zasílané do biomedicínských časopisů.
Klíčová slova:
Montrealský kognitivní test – reliabilita – ekvivalence – vnitřní konzistence – alternativní verze
Authors:
O. Bezdíček 1,2; H. Georgi 2; E. Panenková 3; S. M. Mcclintock 4,5; T. Nikolai 1; E. Růžička 1; M. Kopeček 2,6
Authors‘ workplace:
Department of Neurology, 1st Faculty of Medicine, Charles University and General University Hospital in Prague, Czech Republic
1; National Institute of Mental Health, Klecany, Czech Republic
2; Department of Psychology, Faculty of Arts, Charles University, Prague, Czech Republic
3; Department of Psychiatry, UT Southwestern Medical Center, Dallas, TX, USA
4; Department of Psychiatry and Behavioral Sciences, Duke University School of Medicine, Durham, NC, USA
5; Department of Psychiatry, Third Faculty of Medicine, Charles University, Prague, Czech Republic
6
Published in:
Cesk Slov Neurol N 2019; 82(3): 332-340
Category:
Original Paper
doi:
https://doi.org/10.14735/amcsnn2019332
Overview
Aim: The purpose of this study was to describe the psychometric properties of the Montreal Cognitive Assessment Czech version Standard form (MoCA-SF) and two Alternate Forms (MoCA-AF). There is limited information regarding the test-retest effect and the reliability of the MoCA.
Methods: Seventy cognitively healthy subjects (mean age 50.33 ± 26.47) were assessed in one session with MoCA-SF and MoCA-AF (7.2 and 7.3) in a counterbalanced fashion.
Results: There was no significant difference between the two MoCA forms (Standard vs. 7.3), MoCA-AF 7.2 being slightly more difficult than the latter. Furthermore, in depth, the exploratory analysis revealed differences between the subtests of the MoCA. However, based on the total score all versions showed sound convergent and discriminative validity.
Conclusion: Our data suggest that the two MoCA-SF and MoCA-AF 7.3 forms are equivalent and useful for repeated administration to minimize the test-retest effect.
等价蒙特利尔认知评估的替代形式
目的:
本研究的目的是描述蒙特利尔认知评估捷克版标准形式(MoCA-SF)和两种替代形式(MoCA-AF)的心理测量特性。关于MoCA的重测效果和信度的信息是有限的。
方法:
70名认知健康的受试者(平均年龄50.33±26.47岁)在一次MoCA-SF和MoCA-AF(7.2和7.3)平衡的情况下接受评估。
结果:
MoCA - af 7.2的难度略高于MoCA - af 7.2。进一步,探索性分析揭示了MoCA各子检验之间的差异。然而,从总分来看,所有版本的效度都表现出良好的收敛性和区分性。
结论:
我们的数据表明,MoCA-SF和MoCA-AF 7.3两种剂型是等效的,可重复给药,最大限度地降低复测效果。
关键词:
蒙特利尔认知评估。可靠性。等价性。内部一致性。替代形式
Keywords:
Montreal Cognitive Assessment – Reliability – equivalence – internal consistency – alternate forms
Introduction
There is an emerging need for brief and psychometrically sound screening instruments to measure and document global cognitive abilities in symptomatic predementia phases of neurodegenerative diseases [1–5]. The importance of measuring cognitive performance over short periods necessitates the development of psychometrically sound cognitive instruments that are suitable for test-retest purposes. Consequently, the accuracy of measurement is compromised by measurement error and practice effect, which may alter a true score. Measurement error is one particular source of chance variance that can decrease test-retest reliability [6] and the practice effect is inflation in a true score that suggests artificially improved performance. Generally, there may be a higher propensity for practice effects on repeated testing due to intact cognitive abilities [7–9]. The attenuation of practice effect may be reduced by using a different form, an alternate form, of the test for each assessment session [10]. The alternate form reliability coefficient was originally developed to minimize to the greatest extent possible influence of learning and memory processes on the test-retest assessment of cognitive functioning [11]. To evaluate consistency between form versions, a coefficient of equivalence is computed and reflects the extent to which test forms are interchangeable [12]. The reliability of alternate forms is affected primarily by measurement error related to the test forms [13]. The Montreal Cognitive Assessment (MoCA) is a brief screening test that assesses global cognitive function. Importantly, it has been found useful for the detection of mild cognitive impairment (MCI) and dementia [14] and as such is widely used, available in many languages [15] and recommended as a standard tool for identifying cognitive deficits in patients with neurodegenerative diseases [14,16,17]. The MoCA covers eight cognitive domains in seven subtests including executive functions, visuospatial abilities, short-term memory, language, attention, concentration, working memory and orientation [14]. The MoCA takes approximately 10 to 12 min to complete in healthy subjects [5].
In addition to the original Standard version (7.1), the MoCA has two alternate forms (versions 7.2 and 7.3) [15]. Prior research has showed that both alternate forms yielded equivalent total scores in healthy subjects and patients with MCI or Alzheimer’s disease in the original English version as well as in the German, Italian or French and other language versions [18–23], however, there were also findings regarding the differential difficulty level of the alternate versions in comparison with the corresponding original items [24]. Furthermore, there is limited evidence concerning comprehensive psychometric properties (equivalence, internal consistency) for the MoCA Alternate forms (MoCA-AF) in other languages [18,20,21,25]. Other studies reported only one or two types of reliability for the MoCA Standard forms (MoCA-SF) at a time: test-retest [26–32]; internal consistency [26–28,31,33–35]; reliable change index [9,32,36] and inter-rater reliability for the MoCA clock drawing task only [37].
The reliability of the instrument is a necessary condition for the validity of measurement [13]. As there are limited psychometrically sound cognitive screening instruments in the Czech language, we sought to study the Czech language version of the MoCA-SF and AF. Thus, the aims of the present study were to assess the reliability of MoCA-SF and AF in the Czech version to determine if they were equivalent in healthy subjects; and examine the comprehensive psychometric properties (test-retest reliability, internal consistency and split-half reliability) of the MoCA-SF and AF.
Patients and Methods
Participants
Seventy cognitively healthy Caucasian subjects (46 females) participated in the study (Tab. 1). We recruited all the subjects from university student volunteers or the National Normative Study of Cognitive Determinants of Healthy Ageing study described in detail elsewhere [5]. The exclusion criteria were as follows: no history of brain damage, psychiatric illness, substance or alcohol abuse or any medical illness that affects the brain. None of the participants reported subjective memory complaints or had impaired activities of daily living [38]. The institutional ethics committee and review board approved the study and all the subjects provided their written consent.

Materials
The MoCA-SF: The Czech version of the MoCA was approved and published by the test authors in 2006 (available online) [15]. Since that time, the Czech version has been used in three studies; two focused on Parkinson’s disease [26,39] and the other on Huntington’s disease [34]. The studies reported that the MoCA had high discriminative validity. Additional studies provided normative data in the Czech population and reliable change indices for the MoCA [5,32].
The MoCA-AF (versions 7.2 and 7.3) are available in the English language. We constructed Czech language versions of both MoCA-AF according to the original AF [15]. Our translation resulted in items identical to the English language version, except for certain items in the Language and Memory subtests (see Appendices 1–3). Specifically, two sentences in the language repetition task and five words in the memory task were adapted through the process of translation and back translation to ensure semantic equivalence to the original and the Czech Standard form used. A detailed explanation of the psychometric differences between MoCA-SF English and the Czech version can be found in Kopecek et al [5]. Alterations were created for the phonemic fluency portion of the MoCA between the English and Czech language versions. For the MoCA-AF 7.3 version, the letter ‘B’ for phonemic fluency was replaced with the letter ‘P’ because Czech normative data exist for the letters K and P, which are more suitable regarding the phonological and graphemic system of Czech [40]. The equivalency of ‘K’ and ‘P’ in Czech was confirmed by a previous study [41]. For the English language version of the MoCA-SF, phonemic fluency is assessed with the letter ‘F’ but the letter ‘K’ was used in the Czech MoCA-SF form. In the MoCA-AF 7.2 version, the letter ‘S’ remained the same as in the original because two Czech studies showed similar difficulty for the letter ‘S’ as for the letters ‘K’ and ‘P’ [40,41]. Finally, both MoCA-AF Czech versions were approved by the originators of the MoCA [15].
Procedure
The data collection design for testing the reliability of the MoCA-AF was based on counterbalancing testing order in the single session to eliminate ‘test sophistication effect’ [42]. Randomization was based on permutations of the forms (see Tab. 2).
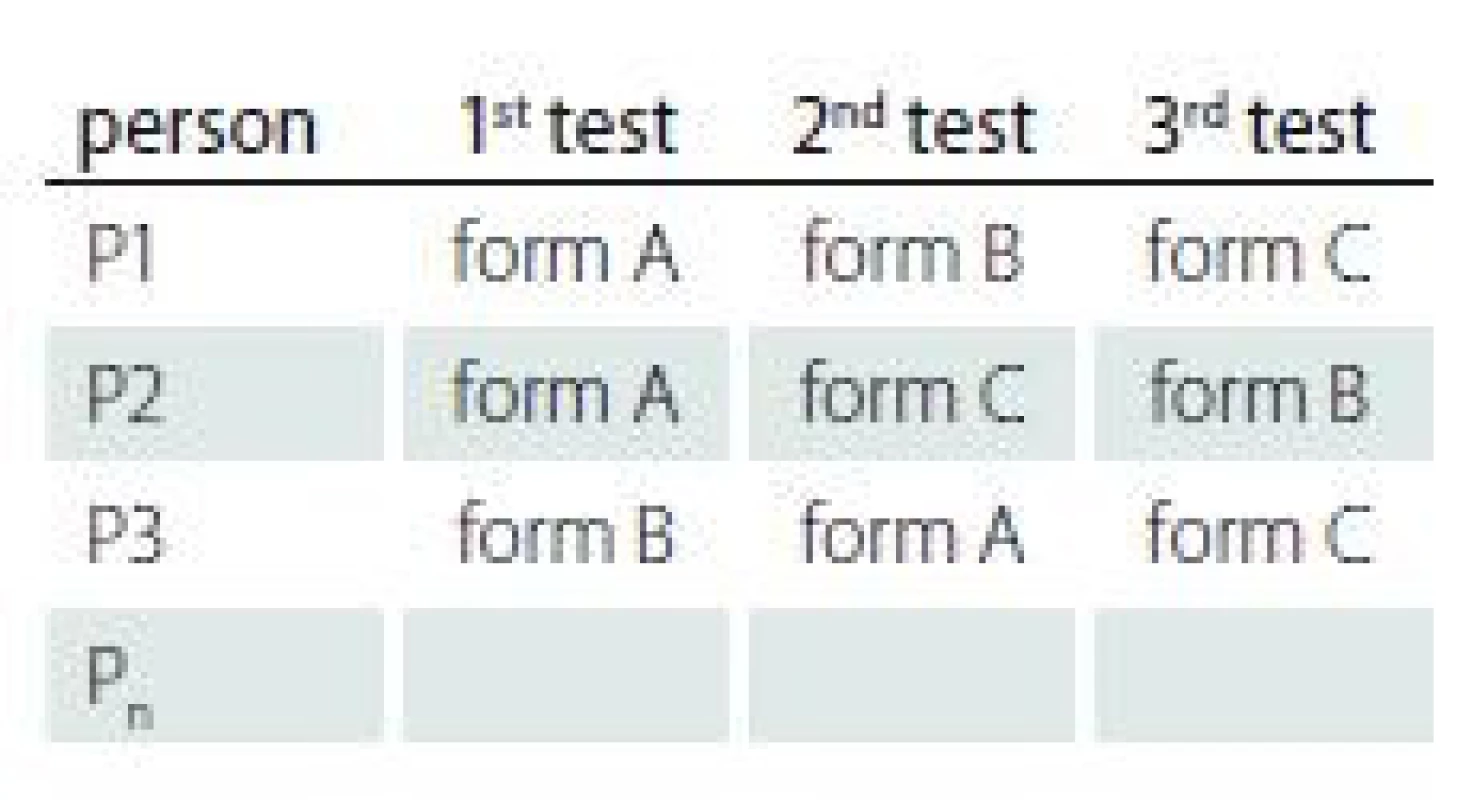
Three MoCA administrations followed one immediately after the other in one session. We did not repeat the Orientation domain (data collected on the same day). Inter-rater reliability was estimated independently by two clinical neuropsychologists (O. B., T. N.). The test-retest reliability was performed only for the MoCA-SF with data collected on 30 young subjects. The time interval between testing time points was on average 42 days (range: 21–58 days). Three subjects did not complete testing at the follow-up time point due to loss of motivation.
Statistical analyses
We assessed for normality of data with the Kolmogorov-Smirnov test and by visual inspection of the Q-Q plot. Correlational analyses for continuous and normally distributed data were carried out with Pearson Product-Moment (Pearson) correlation, while the Spearman Rank correlation method was used for ranked or non-normally distributed data (with the median and interquartile range as descriptive values) and Phi correlation for dichotomous variables. One-way repeated measure analysis of variance (ANOVA) design was applied to evaluate alternate forms equivalence. For between-subtests analysis, the Wilcoxon signed-rank test was used. Significant differences (p < 0.05) were further corrected using the Bonferroni correction for post hoc comparisons to keep the Type I error at 5% overall.
The alternate forms reliability was estimated with Pearson correlation coefficients across the three test administrations. The internal consistency of the MoCA-SF and AF was estimated by Cronbach’s alpha coefficient. Split-half reliability (the correlation of scores between odd/ even subtests) was assessed using the Pearson correlation. Inter-rater reliability was estimated using intra-class correlation coefficients (ICC) using a two-way mixed model consistency definition. To assess the test-retest reliability between scores at baseline and follow-up after one month, we calculated the Pearson correlation coefficient. Finally, the relationships between age, education and practice effect between different MoCA versions were estimated using Pearson correlation coefficients and repeated measures ANOVA. All statistical procedures were performed using SPSS 20.0 for Windows (IBM, Armonk, NY, USA). The significance for all correlations was set at p < 0.05 alpha level.
Results
Alternate forms equivalence
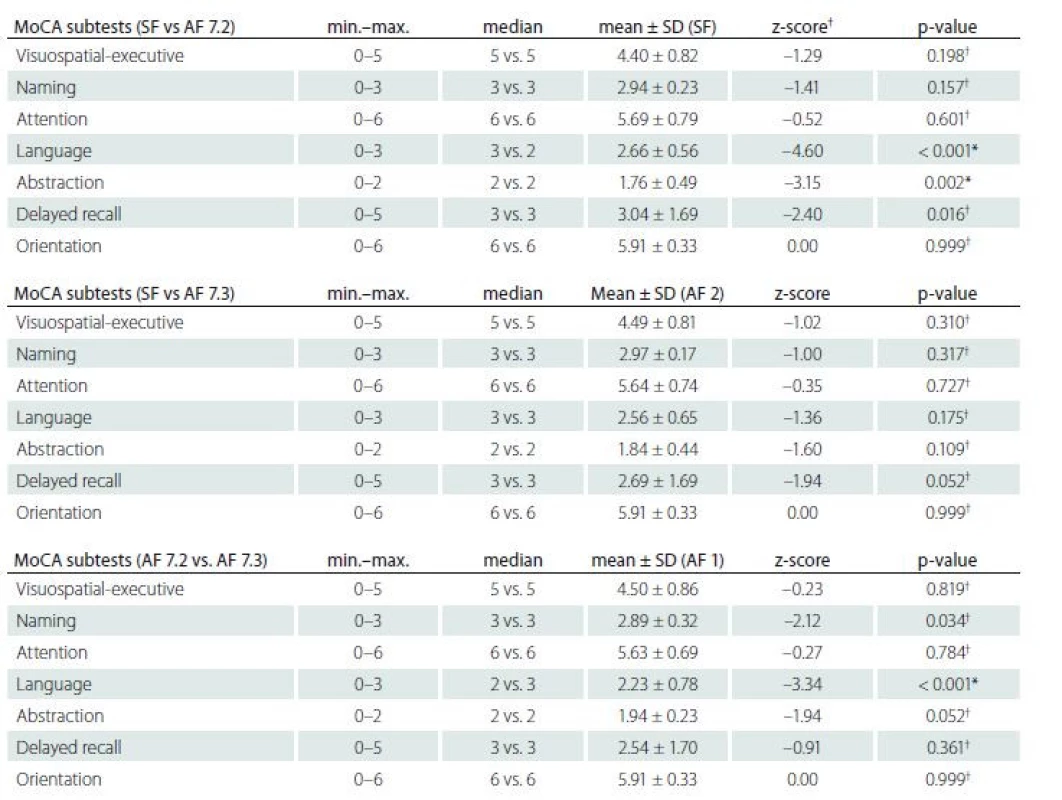
A one-way repeated measures ANOVA was conducted to compare the total scores of MoCA-SF [1] and MoCA-AF (version 7.2 and version 7.3) administered in a counterbalanced order in the single session (see Tab. 2). The means and SD are presented in Tab. 3. There was a non-significant effect for form (Wilks’ Lambda = 0.082, F (2, 68) = 2.59, p = 0.082, multivariate partial eta squared = 0.074). Pairwise comparisons were significant in MoCA-SF vs. 7.2, p = 0.017, however, non-significant with Bonferroni adjustment for multiple comparisons and non-significant in all other comparisons (MoCA-SF vs. MoCA-AF 7.3 and 7.2 vs. 7.3; Tab. 3). The results of ANOVA were replicated by one-way repeated measures ANOVA with age and education as covariates (p = 0.012 for MoCA-SF vs. 7.2), other comparisons being non-significant (after Bonferroni correction as well). A detailed analysis of all seven subtests between the MoCA-SF and AF is presented in Tab. 4. SF and AF 7.2 and 7.3 correlation estimated with Pearson correlation coefficients was 0.77; 0.75; and 0.78, all p΄s = 0.001 (two-tailed).

Other psychometric properties
SF and AF had Cronbach’s alpha coefficient on standardized items of 0.63; 0.64; and 0.54, respectively. Split-half reliability based on the Spearman-Brown prophecy formula for unequal length was 0.46 for SF, 0.54 for AF 2 and 0.58 for AF 3, respectively. The test-retest reliability of the MoCA-SF (range 21–58 days, M = 42, SD = 11.4) using the Pearson correlation coefficient was 0.45 (p = 0.030); we did not find any significant relation between the 21–58 day range and the test or retest MoCA-SF performance (rtest = 0.14, p = 0.555 a rretest= 0.22, p = 0.345). The test-retest ICC of the MoCA was 0.61 (95% CI 0.12–0.83).
Discriminative validity of MoCA Standard and Alternate versions
We split the sample into young (N = 33 subjects) and old (N = 37) age groups (Tab. 1). Comparisons between groups revealed that all MoCA versions showed significant differences (all p΄s < 0.001).
Discussion
The results of the current study show that the MoCA-AF (7.2 and 7.3) in the Czech version are only partially equivalent and interchangeable with the MoCA-SF when applied in healthy subjects. The Alternate forms (especially 7.3) may be used for test--retest purposes in cases of repeated brief assessments. The MoCA-AF 7.2 seems to be more difficult than the MoCA-SF and 7.3 (e. g., the mean difference between the MoCA-SF and MoCA-AF 7.2 total score is 0.67 points and 0.46 points between MoCA--AF 7.2 and 7.3). These results replicate similar findings by Lebedeva et al [24] who selected five items from the MoCA-AF that were included with items from MoCA-SF and used the Rasch model to estimate the difficulty level of the items. However, none of the alternate version items matched the difficulty of their corresponding standard items. Hence for a deeper analysis of MoCA--SF and AF differences between subtests in the present study, we compared all the versions and showed conclusively that there were further differences between MoCA--SF and MoCA-AF 7.2 and even MoCA-AF 7.2 and 7.3 in the Language and Abstraction subtests. However, this discrepancy could not be influenced by changes in the content validity of the MoCA Czech version because all the items in Language use the same syntactic structures as the original. Surprisingly, we surmise that this finding reflects differences in English and Czech syntactical (repeats) and phonological (fluency) systems in which MoCA-AF 7.2 is more difficult than the MoCA-SF and MoCA-AF 7.3 Czech versions. These findings are in accordance with the latest and largest studies on the MoCA-AF differential item difficulty and in general with studies showing a significant language and culture bias in test instruments [21,24,43–45]. Of note is the fact that previous studies on the MoCA-AF did not report on differences between subtests [19,20,23]. All MoCA versions showed medium correlations (> 0.7) and may be considered as highly convergent. The internal consistency of all MoCA forms did not reach a minimum level of 0.7, which is a recommended threshold for Chronbach alpha [46]. The low Cronbach alpha in our study may be due to the small sample size with only healthy adults (and the presence of a ceiling effect) and the number of items on the MoCA. Indeed, psychometric research has established that Cronbach alpha values are dependent on the sample size and the number of items in a scale. For instance, fewer than ten items in a scale (MoCA΄s consistency in the current study was analyzed based on seven subtest scores) and a ceiling effect causing less variance in the data can result in small Cronbach alpha values [46]. As the authors of the original MoCA did not report the alpha values for the alternate versions [18], we were unable to compare and adequately interpret our findings. Comparable studies that used the MoCA-SF reported considerably higher alpha values [14,27,28,31,33,35]. Furthermore, ICC, which can be interpreted as another measure of test-retest reliability was good. Importantly, all MoCA versions were able to differentiate between the young and the old participants in our sample and showed similar levels of high discriminative validity in relation to the expected age effect on the MoCA.
Several important limitations of the present study must be addressed. The current study did not present the accuracy of measurement of the MoCA-AF in clinical samples in comparison with a standard neuropsychological battery. In addition, there is a lack of a medical assessment (imaging and a neurological examination) in the sample. Furthermore, separate normative values for MoCA--AF 7.2 are necessary; hence the normative data for MoCA-SF cannot be applied [5].
In conclusion, the standard MoCA Czech version and the newly constructed MoCA--AF 7.3 are psychometrically sound and were found to be interchangeable and equivalent (irrespective of form) when applied in healthy young and old subjects. However, MoCA-AF 7.2 was significantly more difficult than the latter versions. All MoCA versions have shown similar levels of convergent and discriminative validity. This has important implications for measurement-based care and repeated assessments for neurocognitive screening. Further research is warranted to determine the psychometric properties of the alternate forms in clinical populations with the comprehensive neuropsychological assessment [18–20,22].
The authors would like to express many thanks to the Prague School of Psychosocial Studies for their support. The research was supported with the following grants: Charles University, Progres Q27; Grant Agency of the Czech Republic (‘Cognitive Predictors of Neurodegeneration’, Grant No. 16-01781S); and by LO1611 from the Ministry of Education, Youth and Sport in NPU I.
The authors of the present study signed an official agreement regarding the translation of the MoCA Alternate versions into Czech (7.2 and 7.3) with copyright owner Ziad Nasreddine, M.D.
The authors declare they have no potential conflicts of interest concerning drugs, products, or services used in the study.
The Editorial Board declares that the manuscript met the ICMJE “uniform requirements” for biomedical papers.
Accepted for review: 15. 3. 2019
Accepted for print: 14. 5. 2019
Mgr. Ondřej Bezdíček, Ph.D.
Laboratoř neuropsychologie
J. Diamanta
Neurologická klinika a Centrum klinických neurověd
1. LF UK a VFN
Kateřinská 30, 128 21 Praha 2
Czech Republic
e-mail: ondrej.bezdicek@gmail.com
Sources
1. Cullen B, O‘Neill B, Evans JJ et al. A review of screening tests for cognitive impairment. J Neurol Neurosurg Psychiatry 2007; 78(8): 790–799. doi: 10.1136/ jnnp.2006.095414.
2. Lonie JA, Tierney KM, Ebmeier KP. Screening for mild cognitive impairment: a systematic review. Int J Geriatr Psychiatry 2009; 24(9): 902–915. doi: 10.1002/
gps.2208.
3. Roalf DR, Moberg PJ, Xie SX et al. Comparative accuracies of two common screening instruments for classification of Alzheimer‘s disease, mild cognitive impairment, and healthy aging. Alzheimers Dement 2013; 9(5): 529–537. doi: 10.1016/ j.jalz.2012.10.001.
4. Bezdicek O, Cervenkova M, Moore TM et al. Determining a Short Form Montreal Cognitive Assessment (s-MoCA) Czech version: validity in mild cognitive impairment Parkinson‘s disease and cross-cultural comparison. Assessment 2018 : 1073191118778896. doi: 10.1177/ 1073191118778896.
5. Kopecek M, Stepankova H, Lukavsky J et al. Montreal cognitive assessment (MoCA): normative data for old and very old Czech adults. Appl Neuropsychol Adul 2017; 24(1): 23–29. doi: 10.1080/ 23279095.2015.1065261.
6. Anastasi A, Urbina S. Psychological testing. Upper Saddle River, NJ, USA: Prentice Hall/ Pearson Education 1997.
7. Goldberg TE, Harvey PD, Wesnes KA et al. Practice effects due to serial cognitive assessment: implications for preclinical Alzheimer‘s disease randomized controlled trials. Alzheimers Dement 2015; 1(1): 103–111. doi: 10.1016/ j.dadm.2014.11.003.
8. Basso MR, Bornstein RA, Lang JM. Practice effects on commonly used measures of executive function across twelve months. Clin Neuropsychol 1999; 13(3): 283–292. doi: 10.1076/ clin.13.3.283.1743.
9. Cooley SA, Heaps JM, Bolzenius JD et al. Longitudinal change in performance on the Montreal Cognitive Assessment in older adults. Clin Neuropsychol 2015; 29(6): 824–835. doi: 10.1080/ 13854046.2015.1087596.
10. Beglinger LJ, Gaydos B, Tangphao-Daniels O et al. Practice effects and the use of alternate forms in serial neuropsychological testing. Arch Clin Neuropsychol 2005; 20(4): 517–529. doi: 10.1016/ j.acn.2004.12.003.
11. Calamia M, Markon K, Tranel D. The robust reliability of neuropsychological measures: meta-analyses of test-retest correlations. Clin Neuropsychol 2013; 27(7): 1077–1105. doi: 10.1080/ 13854046.2013.809795.
12. Helmstadter GC. Principles of psychological measurement: East Norwalk, CT, US: Appleton-Century-Crofts 1964.
13. Meyer JP. Reliability. Oxford: Oxford University Press 2010.
14. Nasreddine ZS, Phillips NA, Bedirian V et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 2005; 53(4): 695–699. doi: 10.1111/ j.1532-5415.2005.53221.x.
15. MoCA Montreal – Cognitive Assessment. [online]. Available from URL: https: / / www.mocatest.org/ .
16. Dalrymple-Alford JC, MacAskill MR, Nakas CT et al. The MoCA: well-suited screen for cognitive impairment in Parkinson disease. Neurology 2010; 75(19): 1717–1725. doi: 10.1212/ WNL.0b013e3181fc29c9.
17. Litvan I, Goldman JG, Troster AI et al. Diagnostic criteria for mild cognitive impairment in Parkinson‘s disease: Movement Disorder Society Task Force guidelines. Mov Disord 2012; 27(3): 349–356. doi: 10.1002/ mds.24893.
18. Chertkow H, Nasreddine Z, Johns E et al. The Montreal cognitive assessment (MoCA): Validation of alternate forms and new recommendations for education corrections. Alzheimers Dement 2011; 7 (Suppl 4): S157.
19. Costa AS, Fimm B, Friesen P et al. Alternate-form reliability of the Montreal cognitive assessment screening test in a clinical setting. Dement Geriatr Cogn Disord 2012; 33(6): 379–384. doi: 10.1159/ 000340006.
20. Nasreddine ZS, Patel BB. Validation of Montreal Cognitive Assessment, MoCA, Alternate French Versions. Can J Neurol Sci 2016; 43(5): 665–671. doi: 10.1017/ cjn.2016.273.
21. Siciliano M, Chiorri C, Passaniti C et al. Comparison of alternate and original forms of the Montreal Cognitive Assessment (MoCA): an Italian normative study. Neurol Sci 2019; 40(4): 691–702. doi: 10.1007/ s10072-019-3700-7.
22. Wong A, Yiu S, Nasreddine Z et al. Validity and reliability of two alternate versions of the Montreal Cognitive Assessment (Hong Kong version) for screening of Mild Neurocognitive Disorder. Plos One 2018; 13(5): e0196344. doi: 10.1371/ journal.pone.0196344.
23. Gierus J, Mosiolek A, Koweszko T et al. [The Montreal Cognitive Assessment 7.2 – Polish adaptation and research on equivalency]. Psychiatr Pol 2015; 49(1): 171–179. doi: 10.12740/ PP/ 24748.
24. Lebedeva E, Huang M, Koski L. Comparison of alternate and original items on the Montreal Cognitive Assessment. Can Geriatr J 2016; 19(1): 15–18. doi: 10.5770/ cgj.19.
216.
25. Costa AS, Reich A, Fimm B et al. Evidence of the sensitivity of the MoCA alternate forms in monitoring cognitive change in early Alzheimer‘s disease. Dement Geriatr Cogn Disord 2014; 37(1–2): 95–103. doi: 10.1159/ 000351864.
26. Bezdicek O, Balabanova P, Havrankova P et al.
A Comparison of the Czech version of the Montreal Cognitive Assessment test with the Mini Mental State Examination in identifying cognitive deficits in Parkinson‘s disease. Cesk Slov Neurol N 2010; 73/ 106(2):
150–156.
27. Fujiwara Y, Suzuki H, Yasunaga M et al. Brief screening tool for mild cognitive impairment in older Japanese: validation of the Japanese version of the Montreal Cognitive Assessment. Geriatr Gerontol Int 2010; 10(3): 225–232. doi: 10.1111/ j.1447-0594.2010.00585.x.
28. Lee JY, Dong Woo L, Cho SJ et al. Brief screening for mild cognitive impairment in elderly outpatient clinic: validation of the Korean version of the Montreal Cognitive Assessment. J Geriatr Psychiatry Neurol 2008; 21(2): 104–110. doi: 10.1177/ 0891988708316855.
29. Hu JB, Zhou WH, Hu SH et al. Cross-cultural difference and validation of the Chinese version of Montreal Cognitive Assessment in older adults residing in Eastern China: preliminary findings. Arch Gerontol Geriatr 2013; 56(1): 38–43. doi: 10.1016/ j.archger.2012.05.008.
30. Memoria CM, Yassuda MS, Nakano EY et al. Brief screening for mild cognitive impairment: validation of the Brazilian version of the Montreal cognitive assessment. Int J Geriatr Psychiatry 2013; 28(1): 34–40. doi: 10.1002/ gps.3787.
31. Rahman TT, El Gaafary MM. Montreal Cognitive Assessment Arabic version: reliability and validity prevalence of mild cognitive impairment among elderly attending geriatric clubs in Cairo. Geriatr Gerontol Int 2009; 9(1): 54–61. doi: 10.1111/ j.1447-0594.2008.00509.x.
32. Kopecek M, Bezdicek O, Sulc Z et al. Montreal Cognitive Assessment and Mini-Mental State Examination reliable change indices in healthy older adults. Int J Geriatr Psychiatry 2017; 32(8): 868–875. doi: 10.1002/ gps.4539.
33. Bernstein IH, Lacritz L, Barlow CE et al. Psychometric evaluation of the Montreal Cognitive Assessment (MoCA) in three diverse samples. Clin Neuropsychol 2011; 25(1): 119–126. doi: 10.1080/ 13854046.2010.533
196.
34. Bezdicek O, Majerova V, Novak M et al. Validity of the Montreal Cognitive Assessment in the detection of cognitive dysfunction in Huntington‘s disease. Appl Neuropsychol Adult 2013; 20(1): 33–40. doi: 10.1080/ 09084282.2012.670158.
35. Freitas S, Simoes MR, Maroco J et al. Construct validity of the Montreal Cognitive Assessment (MoCA). J Int Neuropsychol Soc 2012; 18(2): 242–250. doi: 10.1017/ S1355617711001573.
36. Krishnan K, Rossetti H, Hynan LS et al. Changes in Montreal Cognitive Assessment Scores Over Time. Assessment 2017; 24(6): 772–777. doi: 10.1177/ 1073191116654
217.
37. Price CC, Cunningham H, Coronado N et al. Clock drawing in the Montreal Cognitive Assessment: recommendations for dementia assessment. Dement Geriatr Cogn Disord 2011; 31(3): 179–187. doi: 10.1159/ 000324639.
38. Bezdicek O, Stepankova H, Martinec Novakova L et al. Toward the processing speed theory of activities of daily living in healthy aging: normative data of the Functional Activities Questionnaire. Aging Clin Exp
Res 2016; 28(2): 239–247. doi: 10.1007/ s40520-015-0413-5.
39. Bezdicek O, Michalec J, Nikolai T et al. Validity of the Montreal Cognitive Assessment in the Detection of mild cognitive impairment in Parkinson‘s disease. Cesk Slov Neurol N 2014; 77/ 110(1): 47–53.
40. Nikolai T, Stepankova H, Michalec J et al. Tests of verbal fluency, Czech normative study in older patients. Cesk Slov Neurol N 2015; 78/ 111(3): 292–299. doi: 10.14735/ amcsnn2015292.
41. Kopeček M, Kuncová A. Practice effect in Verbal fluency test and Assessment of an alternative version – pilot study. Psychiatrie 2006; 10(4): 211–215.
42. Sacks TL, Clark CR, Pols RG et al. Comparability and stability of performance of six alternate forms of the dodrill-stroop colour-word test. Clin Neuropsychol 1991; 5(3): 220–225. doi: 10.1080/ 13854049108404093.
43. Fernandez AL, Marcopulos BA. A comparison of normative data for the Trail Making Test from several countries: equivalence of norms and considerations for interpretation. Scand J Psychol 2008; 49(3): 239–246. doi: 10.1111/ j.1467-9450.2008.00637.x.
44. Bezdicek O, Moták L, Schretlen DJ et al. Sociocultural and language differences on the Trail Making Test. Arch Assessment Psychol 2016; 6(1): 33–48.
45. van de Vijver F, Tanzer NK. Bias and equivalence in cross-cultural assessment: an overview. Europ Rev Appl Psychol 1997; 47(4): 263–280.
46. Nunnally JC, Bernstein, I. H. Psychometric theory. 3rd ed. New York: McGraw-Hill, Inc. 1994.
Labels
Paediatric neurology Neurosurgery NeurologyArticle was published in
Czech and Slovak Neurology and Neurosurgery

2019 Issue 3
-
All articles in this issue
- Neuromuscular diseases and pregnancy
- Are late complications of Parkinson’s disease really late? YES
- Are late complications of Parkinson’s disease really late? NO
- Are late complications of Parkinson’s disease really late? COMMENT
- Obstructive sleep apnea and cerebral blood flow
- Brief analysis of the frequency of use and spectrum of animal models in stroke research
- Factors affecting the school life of children with epilepsy
- Circadian system disturbances in Huntington’s disease – implications for light therapy
- Experiences with an electrophysiological diagnosis of occupational ulnar nerve lesions at elbow
- Coin in the Hand Test for detection of malingering memory impairment in comparison with mild cognitive impairment and mild dementia in Alzheimer‘s disease
- Neuropathic pain component in patients with myotonic dystrophy type 2 – a pilot study
- Can endarterectomy of the external carotid artery be beneficial? A critical overview
- Inpatient multidisciplinary rehabilitation programme for postural and gait stability in Huntington’s disease – a pilot study
- Optical coherence tomography measurements of the optic nerve head and retina in newly diagnosed idiopathic intracranial hypertension without loss of vision
- Equivalence of Montreal Cognitive Assessment alternate forms
- Frameless and fiducial-less method for deep brain stimulation
- Effect of vacuum-compression therapy for carpal tunnel syndrome as a part of physiotherapy – pilot study
- Anterior choroidal artery aneurysm
- Czech and Slovak Neurology and Neurosurgery
- Journal archive
- Current issue
- About the journal
Most read in this issue
- Coin in the Hand Test for detection of malingering memory impairment in comparison with mild cognitive impairment and mild dementia in Alzheimer‘s disease
- Neuromuscular diseases and pregnancy
- Optical coherence tomography measurements of the optic nerve head and retina in newly diagnosed idiopathic intracranial hypertension without loss of vision
- Effect of vacuum-compression therapy for carpal tunnel syndrome as a part of physiotherapy – pilot study



