Effects of Ditan Tongmai Decoction in combination with acupuncture on post-stroke recovery based on electroencephalogram
Účinky výtažku Ditan Tongmai v kombinaci s akupunkturou na rekonvalescenci po cévní mozkové příhodě hodnocené na základě elektroencefalogramu
Cíl: Cílem studie bylo analyzovat účinky výtažku Ditan Tongmai v kombinaci s akupunkturou při rekonvalescenci po CMP na základě EEG. Metodika: Celkem 110 pacientů ve fázi rekonvalescence po CMP, kteří byli léčeni od února 2019 do listopadu 2021 konvenční západní medikamentózní terapií, bylo ve stejném poměru náhodně zařazeno do kontrolní a kombinační skupiny. Kontrolní skupina byla léčena výtažkem Ditan Tongmai, zatímco v kombinační skupině byl výtažek Ditan Tongmai podáván v kombinaci s akupunkturou. Ve skupinách byly porovnány výsledky léčby, relativní energie frekvenčních pásem EEG, výsledky hodnocení EEG, neurologické a endoteliální funkce. Výsledky: V kombinační skupině bylo celkové procento odpovědí vyšší než v kontrolní skupině (94,5 vs. 81,82 %; p < 0,05). V porovnání s kontrolní skupinou měla kombinační skupina nižší relativní energii frekvenčních pásem β, θ a δ, vyšší relativní energii frekvenčního pásma α a lepší výsledky hodnocení EEG 14 dní po léčbě (p < 0,05). Skóre National Institutes of Health Stroke Scale a sérové hladiny S-100 a N-terminálního natriuretického propeptidu B byly v kombinační skupině nižší než v kontrolní skupině (p < 0,05). V porovnání s kontrolní skupinou byly u kombinační skupiny zaznamenány zvýšené hladiny oxidu dusnatého a calcitonin gene-related peptidu a snížené hladiny endotelinu (p < 0,05). Závěr: V kombinaci s akupunkturou je výtažek Ditan Tongmai, užívaný spolu s konvenční západní medikamentózní terapií, účinný při rekonvalescenci po CMP, přičemž efektivně zlepšuje indikátory EEG a vede k obnovení neurologických a endoteliálních funkcí.
Klíčová slova:
akupunktura – rekonvalescence po cévní mozkové příhodě – výtažek Ditan Tongmai – elektroencefalogram
Authors:
L. Zhang 1*; M. Jin 2*; Q. He 1; X. Liu 1; Y. Hao 1; W. Chen 1
Authors‘ workplace:
The two authors contributed equally to this study.
*; Department of Physical Medicine and Rehabilitation, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou 310000, Zhejiang Province, China
1; Department of Orthopedics, The First Affiliated Hospital of Zhejiang University of Traditional Chinese Medicine, Hangzhou 310000, Zhejiang Province, China
2
Published in:
Cesk Slov Neurol N 2022; 85(4): 306-311
Category:
Original Paper
doi:
https://doi.org/10.48095/cccsnn2022306
Overview
Aim: The aim of the study was to analyze the effects of Ditan Tongmai Decoction in combination with acupuncture on post-stroke recovery based on EEG. Methods: A total of 110 patients with cerebral infarction in the recovery phase treated from February 2019 to November 2021 using the conventional western medicine therapy, were evenly assigned into control and combination groups using a random number table. Control group was treated with Ditan Tongmai Decoction, while Ditan Tongmai Decoction in combination with acupuncture was used for combination group. The treatment outcomes, relative power of EEG frequency bands, EEG evaluation results, neurological function and endothelial function were compared. Results: The overall response rate was higher in combination group than that in control group (94.5 vs. 81.82%; P < 0.05). Compared with control group, combination group had lower relative power of β-frequency band, θ-frequency band and δ-frequency band, higher relative power of α-frequency band and superior EEG evaluation results 14 days after treatment (P < 0.05). The National Institutes of Health Stroke Scale score and serum S-100 and N-terminal pro-B-type natriuretic peptide levels were lower in combination group than those in control group (P < 0.05). Compared with control group, combination group had elevated levels of nitric oxide and calcitonin gene-related peptide and reduced endothelin level (P < 0.05). Conclusion: Ditan Tongmai Decoction in combination with acupuncture used on top of the conventional western medicine therapy, is effective in post-stroke recovery after ischemic stroke, which is capable of efficiently improving EEG indicators and conducive to the recovery of neurological and endothelial functions.
Keywords:
Acupuncture – post-stroke recovery – Ditan Tongmai Decoction – electroencephalogram
Introduction
As the most complicated and crucial organ of the human body, the brain has about 14 billion nerves or neuron cells therein. The neuronal discharge activity in the cerebral cortex is indispensable for advanced activities such as language, analysis, logic, cognition and thinking in human beings [1]. Brain function has been explored basically by virtue of neuroanatomy in the past. However, the secret of brain function mostly lies on the neuronal discharge activity in the cerebral cortex, so detection and analysis of such activity, i.e., neurological function examination, are necessary to deeply understand brain function. Nevertheless, the examination is still under exploration [2]. EEG is the most extensively applied examination for monitoring brain function in clinics, and is able to record spontaneous and rhythmic electrical activity of brain cells, directly mirroring brain metabolism and prompting information about brain dysfunction. Hence, EEG is usually used to diagnose brain dysfunction [3]. Cerebral infarction is a disease of cerebral cortex damage most often diagnosed in middle-aged and elderly population, which has high morbidity, disability and fatality rates and brings a huge burden and suffering to the family of patients [4]. In China, increasingly more people suffer from cerebral infartion yearly due to the intensifying aging of the population [5]. The recovery of patients with cerebral infarction is determined mostly based on anatomy including the disappearance or shrinkage of lesions in the past, with few objective indicators for neurological examination, and there are few studies on the evaluation of the recovery of cerebral infarction using EEG.
This study aimed to assess the effects of Ditan Tongmai Decoction in combination with acupuncture on post-stroke recovery based on EEG.
Patients and methods
General information
A total of 110 patients with cerebral infarction in the recovery phase treated in our hospital from February 2019 to November 2021 were evenly assigned into the control group and combination group using a random number table.
Inclusion criteria
The inclusion criteria were set as follows: patients who met the diagnostic criteria in the Chinese Stroke Association Guidelines for Clinical Management of Cerebrovascular Disorders [6] and were definitely diagnosed with such a disease by imaging examination, those aged below 80 years old, those in the recovery phase, i.e., a disease course of 2 weeks to 6 months, those with National Institutes of Health Stroke Scale (NIHSS) score ≤ 22 points, and those who voluntarily signed the informed consent.
The exclusion criteria involved: patients with cerebral hemorrhage, psychiatric disorders, malignant tumors of vital organs, or hematological or nervous system diseases.
Methods
Conventional western medicine treatment (oral administration with simvastatin 20 mg once/day; oral administration with enteric-coated aspirin tablet 100 mg once/day; supplemented with drugs for stabilizing blood pressure and glucose levels) was employed for both groups. On this basis, the control group was given Ditan Tongmai Decoction. The prescriptions were as follows: Radix paeoniae rubra, Pinellia ternata, Caulis bambusae in taeniam, Gastrodiae elata, Radix angelicae sinensis, and Ligusticum chuanxiong (10 g of each); Arisaema cure bile, Radix cyathulae, Eucommia ulmoides, Taxillus chinensis, Acorus tatarinowii, Concha haliotidis, and Poria cocos (15 g of each); Ramulus uncariae cum uncis (20 g); flowers Carthami, Pericarpium citri reticulatae, and peach kernel (6 g of each); and raw licorice root (3 g). All these medicinal ingredients were decocted in water for the oral dose in the morning and in the evening, with 100 mL/dose, and for 14 consecutive days.
As mentioned above, Ditan Tongmai Decoction combined with acupuncture was applied for the combination group on the basis of western medicine treatment. The major acupoints for acupuncture included Fengchi, Sishencong, Taiyang, Renzhong, Baihui, and Tian Sanzhen, and the auxiliary acupoints were Shousanli, Tongli, Sanyinjiao, Quchi, Yanglingquan, Kunlun and so on. Specifically, 0.5-cun filiform needles were adopted for acupuncture at the Sishencong and Renzhong acupoints with a depth of 0.3–0.4 cun; 2.5-cun filiform needles were utilized for acupuncture at the Fengchi acupoint with a depth of 2 cun; and 2.0-cun filiform needles were employed for acupuncture at other acupoints with a depth of 1.0–1.5 cun. The needles were kept at all acupoints for 20 min. The acupuncture was performed continuously for 14 days, once a day.
Evaluation indices
Clinical efficacy was assessed according to the variation of NIHSS score before and after treatment: a decrease of no less than 91% in the NIHSS score and no disability after treatment suggested healing, a decrease of 45–90% in the NIHSS score and grade 1–3 disability after treatment was considered markedly effective; a decrease of 17–44% in the NIHSS score and grade 1–3 disability after treatment was considered effective; and not meeting the above criteria indicated ineffective. Overall response rate = cured rate + markedly effective rate + effective rate.
The relative power of EEG frequency bands was determined as follows: the brain electrical activity of patients in both groups was detected using a Nuocheng video EEG (Huanxi Medical Technology Co., Ltd., Guangzhou, China) before treatment and at 14 days after treatment, with the electrode placed as per the international 10–20 system, and two ear electrodes as the reference electrode. The brain electrical activity of patients was monitored in the lying position, awake and in quiet state for 2 h. EEG stable for 30 s and formed in the frequency domain of 0.5–30 Hz was selected, which covered the a-frequency band (8.0–13.9 Hz), b-frequency band (14.0–30.0 Hz), θ-frequency band (4.0–7.9 Hz) and d-frequency band (1.0–3.9 Hz). The power was obtained by analog-to-digital fast Fourier transform. The relative power was calculated by the following equation: relative power of a certain frequency band = power of this frequency band / sum of the powers of all frequency bands.
The EEG evaluation results of patients in both groups before and after treatment were compared, which were classified into borderline state, mild abnormality, moderate abnormality, and severe abnormality. Mild abnormality: the a-rhythm is unstable, the patient has no obvious fluctuations when opening the eyes, the b-wave has a high amplitude, and the activity of Q-wave increases in each area; it can also be manifested as an increase in the main activity area and considerable fluctuation of the Q-wave during excessive arousal. Moderate abnormality: the a-rhythm disappears or becomes markedly asymmetrical, the Q-wave activity is diffuse or paroxysmal, and high-amplitude d-wave appears after hyperventilation. Severe abnormality: the activities of d - and Q-waves are diffuse, and the a-wave slows down or even disappears. The d-wave has paroxysmal activity or is accompanied by sharp waves or high-amplitude spike waves.
Neurological function was examined as follows: 5 mL of fasting venous blood was collected from patients in both groups before treatment and at 14 days after treatment and was then centrifuged, followed by collection of the supernatant for detection. The time-resolved fluorescence immunoassay and immunoradiometric assay were conducted to measure the levels of S-100 and N-terminal pro-B-type natriuretic peptide (NT-proBNP), respectively. NIHSS was adopted for the assessment of neurological deficits.
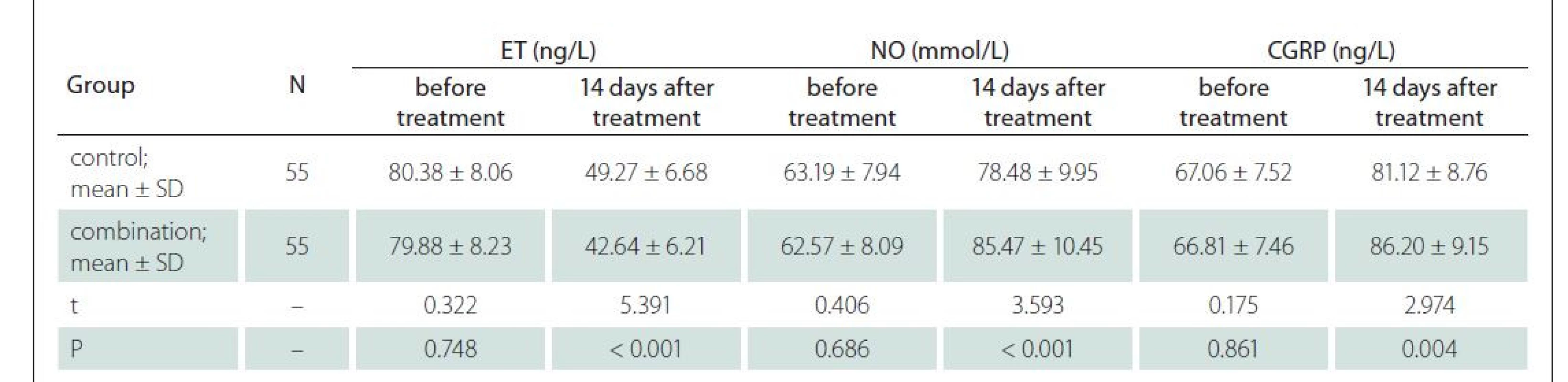
The determination of endothelial function was carried out. Specifically, the blood was collected and processed as above. The endothelial function of patients in both groups, including calcitonin gene-related peptide (CGRP), nitric oxide (NO), and endothelin (ET), was determined through the double antibody sandwich enzyme-linked immunosorbent assay.
Statistical analysis
Statistical analysis was conducted using SPSS 24.0 software (IBM, Armonk, NY, USA). Measurement data were expressed as mean ± standard deviation (SD) and subjected to the t-test. Numerical data were expressed as rate and subjected to the c2 test. P < 0.05 suggested a statistically significant difference.
Results
Clinical and demographic characteristics
In the control group, there were 29 males and 26 females aged 50–77 years, with a mean age of 62.29 ± 5.25 years. The course of the disease was from 15 days to 5 months, with an average of 2.10 ± 0.26 months. The NIHSS score was 15.10 ± 2.37 points. Among them, 29 cases developed lacunar cerebral infarction, and 26 cases suffered from cerebral thrombosis, with 23 cases complicated by arterial hypertension, 20 cases complicated by diabetes mellitus, and 13 cases complicated by hyperlipidemia. In the combination group, there were 28 males and 27 females aged 50–77 years old, with a mean age of 64.65 ± 5.11 years. The course of disease was from 15 days to 6 months, with an average of 2.18 ± 0.24 months. The NIHSS score was 14.95 ± 2.45 points. Among them, 30 cases had lacunar cerebral infarction, and 25 cases developed cerebral thrombosis, with 21 cases complicated by arterial hypertension, 21 cases complicated by diabetes mellitus, and 12 cases complicated by hyperlipidemia. The two groups had similar and comparable general data (P > 0.05).
Clinical treatment outcomes
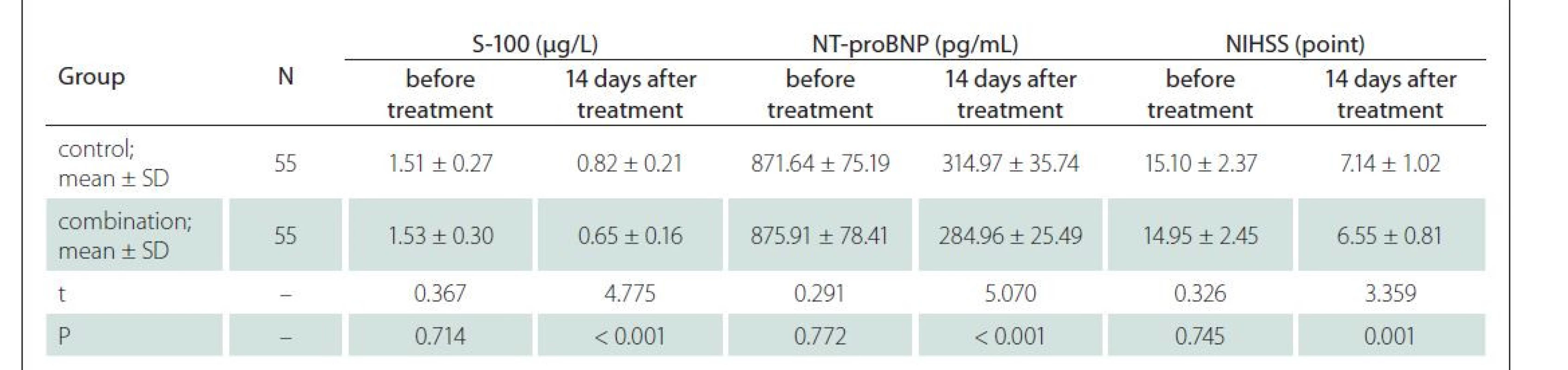
The overall response rate of the combination group was significantly higher than that of the control group (94.55 vs. 81.82%, P < 0.05) (Tab. 1).
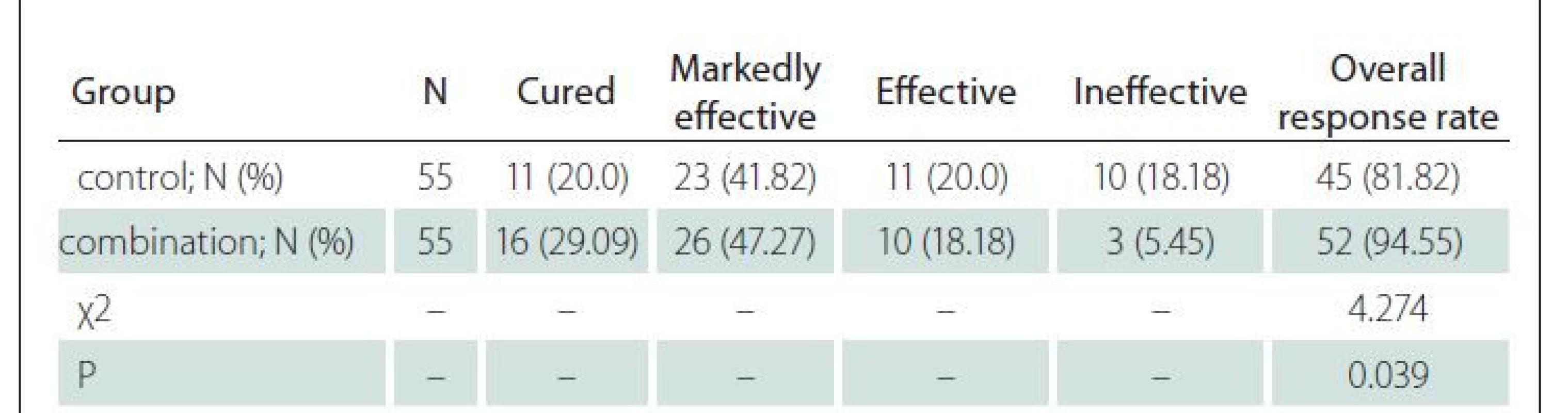
Relative powers of EEG frequency bands
Before treatment, the two groups had similar relative powers of all frequency bands (P > 0.05). The combination group had lower relative powers of the b-frequency band, θ-frequency band and d-frequency band and higher relative power of a-frequency band than those of the control group 14 days after treatment (P < 0.05) (Tab. 2).
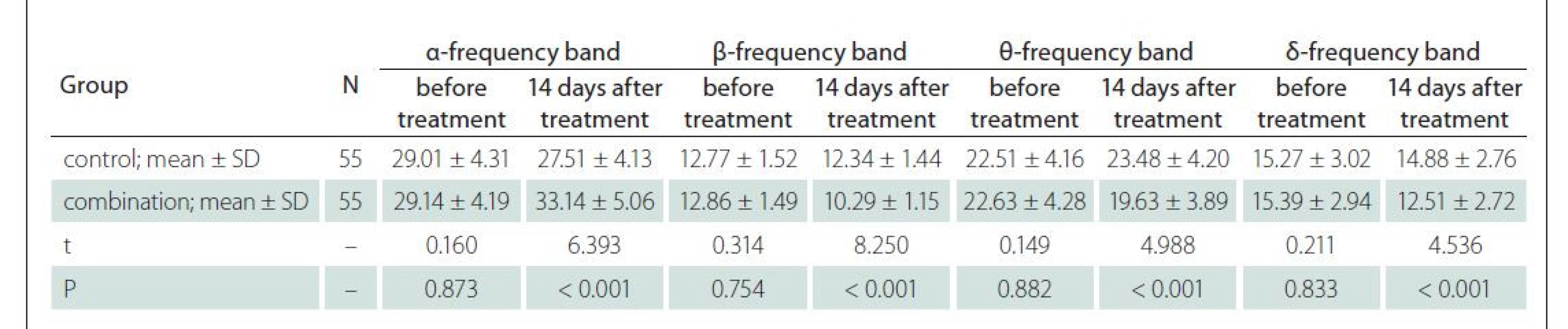
EEG evaluation results
The combination group had superior EEG evaluation results compared to those of the control group 14 days after treatment (P < 0.05) (Tab. 3).
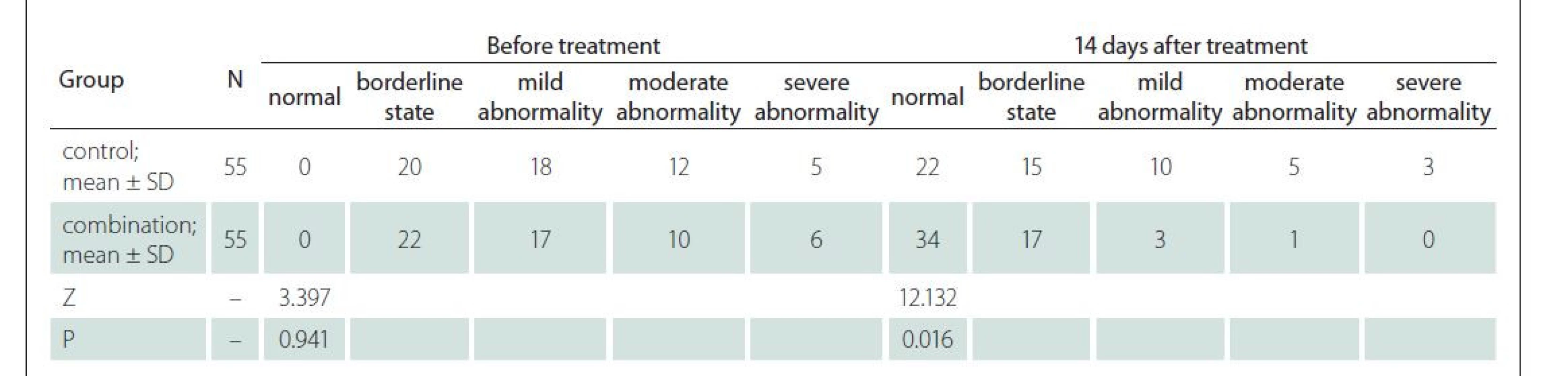
Neurological function
Before treatment, the two groups had similar NIHSS scores and serum S-100 and NT-proBNP levels (P > 0.05). The NIHSS scores and serum S-100 and NT-proBNP levels were lower in the combination group than those in the control group 14 days after treatment (P < 0.05) (Tab. 4).
Endothelial function
Before treatment, the two groups had similar NO, CGRP and ET levels (P > 0.05). Compared with the control group, the combination group had elevated levels of NO and CGRP and reduced ET levels 14 days after treatment (P < 0.05) (Tab. 5).
Discussion
Electroencephalography is able to respond to the brain bioelectric activity of the cerebral cortex, and EEG refers to a graphical record by magnifying the spontaneous bioelectrical potential of the brain through electronic instruments on the scalp. The electrical activity of the brain cell mass recorded through electrodes can be used to evaluate the functional state of the cerebral cortex, which is mostly utilized to assess the condition of patients with cerebral cortex damage such as disturbance of consciousness. EEG has become a vital indicator for monitoring nerve function and function damage of the brain due to advantages of non-invasiveness and convenience [7,8]. The structural damage of the brain can be detected by MRI, CT and other methods, but these methods cannot detect the changes in brain function, which can be made up by EEG. The degree of abnormalities on EEG has a close association with the location of lesions and the accuracy of localization. Inactivated brain cells in the infarcted area have no electrical activity, while damaged ones display abnormal electrical activity, and the variations in such activity exhibit gradient change with the distance from lesions [9]. The specific location and scope of lesions cannot be directly indicated by EEG, but EEG can sensitively reflect changes in brain function, rendering a reference for the assessment of the efficacy and prognosis of cerebral infarction [10].
The electrical activity of the brain is mainly produced in the large pyramidal cells of the cerebral cortex. Some scholars have pointed out that d-activity is the major electrical activity of the brain in cerebral infarction regions, while in the case of cytotoxic edema, θ-activity is the major electrical activity of the brain [11]. Imbalanced brain cell metabolism, decreased cerebral blood flow, and abnormal activity of neurons in patients with cerebral infarction can be sensitively detected by EEG, which is a prerequisite for the assessment of the recovery of cerebral infarction by EEG [12]. Abnormal d-activity is closely correlated with the blockage of the nerve conduction pathway of electrical activity in necrotic tissues, which can denote the severity of cerebral infarction, and abnormal θ-activity can mirror the severity of cell edema, which is a pathological premise for the assessment of the recovery of cerebral infarction by EEG [13]. The a-frequency band and b-frequency band are fast wave frequency bands. The b-wave has close correlation with cognitive function, and its decrease indicates mild cognitive impairment, whereas the a-wave is the basic waveform of EEG, and its decrease suggests an inhibited cerebral cortex [14,15]. In this study, it was found that at 14 days after treatment, the combination group displayed lower relative power of the b-frequency band, θ-frequency band and d-frequency band, higher relative power of the a-frequency band and superior EEG evaluation results in comparison with the control group, signifying that Ditan Tongmai Decoction in combination with acupuncture is able to effectively improve the EEG indicators of patients with cerebral infarction in the recovery phase and is conducive to the recovery of brain function.
The brain is highly sensitive to ischemia and hypoxia, leading to neurological impairment and inflammatory reactions [16]. As to the treatment of cerebral infarction, conventional western medicine therapy mainly focuses on thrombolysis, mechanical recanalization, anticoagulation, administration of antiplatelets, and reduction of blood pressure, hyperlipidemia, and blood glucose. As traditional Chinese medicine (TCM) rises in popularity in recent years, various TCM therapies including TCM and acupuncture have achieved satisfactory results in the treatment of cerebral infarction. The results of this study revealed that, on top of the conventional western medicine therapy, the combination group displayed a higher overall response rate than the control group, and the NIHSS scores and serum S-100 and NT-proBNP levels were lower in the combination group than those in the control group at 14 days after treatment, implying that Ditan Tongmai Decoction in combination with acupuncture is able to effectively improve the efficacy in patients with cerebral infarction in the recovery phase and improve the EEG indicators and nervous function in comparison with the use of Ditan Tongmai Decoction only. The reason is that Acorus tatarinowii in Ditan Tongmai Decoction can promote blood circulation and regulate qi, and remove phlegm and induce resuscitation, which can cure epilepsy, phlegm syncope, etc. Radix angelicae sinensis and Ligusticum chuanxiong have the effect of dispelling blood stasis and promoting blood circulation, and Panax notoginseng has the effect of dispersing blood stasis and subsiding swellings. Ramulus uncariae cum uncis acts on the liver and pericardium meridians and is capable of calming the liver and clearing heat, as well as calming endogenous wind and arresting convulsion. Pinellia ternata has the effect of removing dampness to reduce phlegm, as well as dissolving lumps and resolving masses. The combination of various medicines exerts the function of removing blood stasis and promoting blood circulation, as well as dispelling wind and removing obstruction in the meridians [17]. In this study, acupuncture at such acupoints as Baihui, Sishencong, Renzhong and Fengchi is able to effectively relieve cerebral spasm, thus improving cerebral hemodynamic disorders. Simultaneous acupuncture at many acupoints can form a system that positively affects the circulatory system, contributing to the improvement of hemodynamics, and even reversing the progression of cerebral infarction [18]. Acupuncture at related acupoints of the brain is also capable of modulating the permeability of the mitochondrial membrane of neuron cells and repressing the synthesis and production of free radicals, thereby protecting neuron cells [19]. It was discovered in this study that in contrast with the control group, the combination group had elevated levels of NO and CGRP and a declined ET levels at 14 days after treatment, demonstrating that the combination of Ditan Tongmai Decoction and acupuncture can effectively improve vascular endothelial function in patients undergoing cerebral infarction recovery. It is because acupuncture at the relevant acupoints of the brain can stimulate the anterior and posterior circulation of the brain, promote the recovery of collateral circulation, and regulate the level of neurotransmitters to some extent, improving blood viscosity and thus protecting endothelial function to a certain extent [20]. Additionally, it has been proved by modern pharmacology that Ligusticum chuanxiong and peach kernel in Ditan Tongmai Decoction can resist platelet aggregation, reduce blood viscosity and resist thrombus formation. Acupuncture in combination with Ditan Tongmai Decoction can protect the endothelial function of patients [21].
In conclusion, Ditan Tongmai Decoction in combination with acupuncture, on top of the conventional western medicine therapy, is effective for post-stroke recovery after ischemic stroke, which is capable of efficiently improving EEG indicators and conducive to the recovery of neurological and endothelial functions.
Ethical principles
The entire study was conducted in accordance with the Helsinki Declaration of 1975 (as revised in 2004 and 2008). The ethic committee approval number is “Sir Run Run Shaw Hospital 2022-0217” (February 4th, 2019). Written informed consent has been obtained from all patients.
Conflict of interest
The authors declare they have no potential conflicts of interest concerning drugs, products, or services used in the study.
Wenjun Chen
Department of Physical Medicine
and Rehabilitation
Sir Run Run Shaw Hospital
Zhejiang University School
of Medicine
Hangzhou 310000
Zhejiang Province
China
e-mail: 3191033@zju.edu.cn
Accepted for review: 24. 3. 2022
Accepted for print: 21. 7. 2022
Sources
1. Wu D, Liu X, Gadhoumi K et al. Causal relationship between neuronal activity and cerebral hemodynamics in patients with ischemic stroke. J Neural Eng 2020; 17 (2): 026006. doi: 10.1088/1741-2552/ab75af.
2. Ajčević M, Furlanis G, Naccarato M et al. Hyper-acute EEG alterations predict functional and morphological outcomes in thrombolysis-treated ischemic stroke: a wireless EEG study. Med Biol Eng Comput 2021; 59 (1): 121–129. doi: 10.1007/s11517-020-02280-z.
3. Cao Y, Song X, Wang L et al. Transcranial doppler combined with quantitative electroencephalography brain function monitoring for estimating the prognosis of patients with posterior circulation cerebral infarction. Front Neurol 2021; 12 : 600985. doi: 10.3389/fneur. 2021.600985.
4. Elmali AD, Ekizoglu E, Ciftci I et al. Periodic electroclinical seizures following an ischemic stroke revealed by continuous-EEG. Epilepsy Behav Rep 2021; 15 : 100428. doi: 10.1016/j.ebr.2021.100428.
5. Wilkinson CM, Burrell JI, Kuziek JWP et al. Predicting stroke severity with a 3-min recording from the Muse portable EEG system for rapid diagnosis of stroke. Sci Rep 2020; 10 (1): 18465. doi: 10.1038/s41598-020-75 379-w.
6. Liu L, Chen W, Zhou H et al. Chinese Stroke Association guidelines for clinical management of cerebrovascular disorders: executive summary and 2019 update of clinical management of ischaemic cerebrovascular diseases. Stroke Vasc Neurol 2020; 5 (2): 159–176. doi: 10.1136/svn-2020-000378.
7. Saga N, Doi A, Oda T et al. Elucidation of EEG characteristics of fuzzy reasoning-based heuristic BCI and its application to patient with brain infarction. Front Neurorobot 2021; 14 : 607706. doi: 10.3389/fnbot.2020.607 706.
8. Manganotti P, Furlanis G, Ajčević M et al. CT perfusion and EEG patterns in patients with acute isolated aphasia in seizure-related stroke mimics. Seizure 2019; 71 : 110–115. doi: 10.1016/j.seizure.2019.07.005.
9. Erani F, Zolotova N, Vanderschelden B et al. Electroencephalography might improve diagnosis of acute stroke and large vessel occlusion. Stroke 2020; 51 (11): 3361–3365. doi: 10.1161/STROKEAHA.120.030150.
10. Ajčević M, Furlanis G, Miladinović A et al. Early EEG alterations correlate with CTP hypoperfused volumes and neurological deficit: a wireless EEG study in hyper-acute ischemic stroke. Ann Biomed Eng 2021; 49 (9): 2150–2158. doi: 10.1007/s10439-021-02735-w.
11. Shreve L, Kaur A, Vo C et al. Electroencephalography measures are useful for identifying large acute ischemic stroke in the emergency department. J Stroke Cerebrovasc Dis 2019; 28 (8): 2280–2286. doi: 10.1016/j.jstrokecerebrovasdis.2019.05.019.
12. Kruse A, Suica Z, Taeymans J et al. Effect of brain--computer interface training based on non-invasive electroencephalography using motor imagery on functional recovery after stroke – a systematic review and meta-analysis. BMC Neurol 2020; 20 (1): 385. doi: 10.1186/s12883-020-01960-5.
13. Saga N, Doi A, Oda T et al. Elucidation of EEG characteristics of fuzzy reasoning-based heuristic BCI and its application to patient with brain infarction. Front Neurorobot 2021; 14 : 607706. doi: 10.3389/fnbot.2020.607 706.
14. Liu X, Pu Y, Wu D et al. Cross-frequency coupling between cerebral blood flow velocity and EEG in ischemic stroke patients with large vessel occlusion. Front Neurol 2019; 10 : 194. doi: 10.3389/fneur.2019.00194.
15. Tian J, Liu LD, Zhou Y et al. The change index of quantitative electroencephalography for evaluating the prognosis of large hemispheric infarction. J Integr Neurosci 2021; 20 (2): 341–347. doi: 10.31083/j.jin2002 033.
16. Zhou D, Xie L, Wang Y et al. Clinical efficacy of tonic traditional Chinese medicine injection on acute cerebral infarction: a Bayesian network meta-analysis. Evid Based Complement Alternat Med 2020; 2020 : 8318792. doi: 10.1155/2020/8318792.
17. Liu R, Yu X, Zhang L et al. Computed tomography (CT) imaging evaluation of integrated traditional Chinese medicine cooperative therapy in treating acute cerebral infarction: a randomized controlled trial. Medicine (Baltimore) 2020; 99 (18): e19998. doi: 10.1097/MD.00 00000000019998.
18. Yan J, Dong Y, Niu L et al. Clinical effect of Chinese herbal medicine for removing blood stasis combined with acupuncture on sequelae of cerebral infarction. Am J Transl Res 2021; 13 (9): 10843–10849.
19. Zhang T, Zhao J, Li X et al. Chinese Stroke Association guidelines for clinical management of cerebrovascular disorders: executive summary and 2019 update of clinical management of stroke rehabilitation. Stroke Vasc Neurol 2020; 5 (3): 250–259. doi: 10.1136/svn-2019-000321.
20. Jia H, He J, Zhao L et al. Combination of stem cell therapy and acupuncture to treat ischemic stroke: a prospective review. Stem Cell Res Ther 2022; 13 (1): 87. doi: 10.1186/s13287-022-02761-y.
21. Su XT, Wang L, Ma SM et al. Mechanisms of acupuncture in the regulation of oxidative stress in treating ischemic stroke. Oxid Med Cell Longev 2020; 2020 : 7875396. doi: 10.1155/2020/7875396.
Labels
Paediatric neurology Neurosurgery NeurologyArticle was published in
Czech and Slovak Neurology and Neurosurgery

2022 Issue 4
-
All articles in this issue
- Editorial
- Dabigatran pharmacogenetics and secondary prevention of ischemic stroke
- Validation of questionnaire for evaluation of ischemic stroke sequels – the Czech version of Stroke Impact Scale 3.0
- Telemedical assessments by remote versions of ALBA, POBAV and ACE-III tests
- Heart rate variability analysis during head-up tilt testing in diagnostics of reflex syncope – review of problems and our experiences
- Delirium management in neurointensive care in the Czech Republic – a survey
- Pathological magnetic resonance imaging findings in myelin oligodendrocyte glycoprotein antibody-associated disease
- Zemřel profesor Zdeněk Mraček
- Effects of Ditan Tongmai Decoction in combination with acupuncture on post-stroke recovery based on electroencephalogram
- Rituximab treatment of post-COVID-19 development of acute-onset chronic inflammatory demyelinating polyneuropathy
- Czech and Slovak Neurology and Neurosurgery
- Journal archive
- Current issue
- About the journal
Most read in this issue
- Validation of questionnaire for evaluation of ischemic stroke sequels – the Czech version of Stroke Impact Scale 3.0
- Telemedical assessments by remote versions of ALBA, POBAV and ACE-III tests
- Pathological magnetic resonance imaging findings in myelin oligodendrocyte glycoprotein antibody-associated disease
- Delirium management in neurointensive care in the Czech Republic – a survey
